Motor Proteins Move on Metal
Proteins are very large molecules that do most of the work in biology, and in recent years biophysicists have begun to detect the action of single protein molecules at work, rather than measuring only ensemble properties. In 1995, a Japanese group observed the first fluorescence from single working proteins in water, and in the 18 May PRL the team shows that a metal surface can enhance the fluorescence and allow for a new class of experiments involving proteins on metal surfaces.
Biophysicists often use fluorescence–the light emitted by a molecule after being excited by light–to study biomolecules because it can probe chemical reactions and conformational changes with high sensitivity. But the fluorescence from a single molecule in water is normally outshined by incident light scattered by water molecules, luminescence of dust in the solution, and other effects. Researchers use a variety of tricks to illuminate only a very small volume of water and reduce the background light. The Japanese team, led by Toshio Yanagida of Osaka University, used total internal reflection: The exciting laser reflects from the far surface of a quartz slide at a glancing angle, generating an evanescent wave that penetrates just far enough on the other side to illuminate protein molecules at the surface. They also took pains to use nonfluorescent optics and extremely narrow-band optical filters.
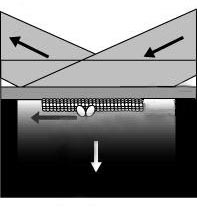
In their latest experiments, the group coated the slide with a thin metal film that enhanced the fluorescence through surface plasmons–electromagnetic waves that travel on a metal surface. The plasmons provided more intense excitation of the fluorescence without the ill-effects of turning up the laser. They observed single, fluorescently labeled kinesin molecules moving many microns along “tracks” of the microtubule biopolymer, just as they do when shuttling cargo inside of cells. To see the action of myosin, the motor protein that makes muscles move, they inverted the geometry, sticking many motors to the surface, and watching the biopolymer actin snake along above them, as the “bucket brigade” of motors moved it. In both cases, all of the molecules had fluorescent labels.
Although proteins often become congealed and disordered in high concentrations, several researchers have shown that proteins on a metal surface can become well organized into arrays, as sulfur and other protein atoms bond with the surface. Yanagida hopes to study these well-organized proteins using his fluorescence technique, but he first had to prove the method would work on a metal. Since metal atoms normally “quench,” nearby fluorescence–inhibit it by absorbing the energy of the excited state– Yanagida says he was surprised the fluorescence was unaffected in this case.
Shimon Weiss, of the Lawrence Berkeley National Laboratory, says the experiment is “very interesting,” but he would like to see data explicitly demonstrating an increased signal-to-background ratio resulting from the metal surface, compared with bare quartz. On the other hand, he agrees that many experiments involving proteins on metal surfaces could benefit from this new type of microscope.


