All Twisted Up
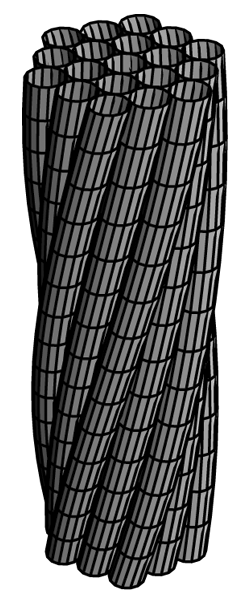
Patients with sickle cell disease have mutant hemoglobin proteins that form long, stiff fibers inside red blood cells. In the 28 March PRL, researchers propose a mathematical model to explain the persistent stability of these deadly fibers. The theory suggests that an inherent “twistiness” in the strands that make up the fibers could be the key to their durability and possibly to new treatments.
Red blood cells supply oxygen to the body using their cargo of hemoglobin, a protein that can capture and release oxygen. Hemoglobin molecules normally float freely in the cell, but sickle cell patients have a mutated, “sticky,” form of hemoglobin that tends to clump together into long fibers. The stiff fibers form a scaffolding that distorts the cells into their namesake “sickle” shape, so they jam up trying to pass through small blood vessels. The traffic jams deprive vital organs of oxygen, so patients end up with anemia, jaundice, major organ damage, and many other maladies.
A sickle hemoglobin fiber can be made up of anywhere from 14 to more than 400 individual strands of hemoglobin molecules linked into long chains. Matthew Turner, of the University of Warwick in the UK, wondered why these strands tend to clump together into long, stiff, fibers rather than compact crystals, which would be less harmful. “A scaffolding made of the rigid fibers is much worse than a couple little sugar-cube-like crystals floating around,” Turner says. So he and his colleagues constructed a mathematical model.
The team’s equations start with the trade-offs that exist in any material as it tries to find the shape with the least overall stress. The forces at work include bending and stretching, and for hemoglobin strands, there is also a propensity to stick together. This stickiness would normally make a thick, compact crystal more stable than a thin fiber, Turner explains, because a crystal maximizes the contact area of the protein with itself. But for sickle hemoglobin, fibers are more stable. To favor fibers, the equations needed to include the fact that the individual strands of molecules are inherently “twisty.” They behave like the coiled wire that attaches a telephone to its handset, apparently because the molecules link up in a way that favors twisting. The strands wrap around one another like threads of rope to form the fibers. In their paper, the team shows that their model’s predictions for two of the mechanical properties of fibers agree with experiments.
Turner says that the model suggests a possible treatment for sickle cell disease. Gene therapy could introduce a hemoglobin mutant that formed less-twisty individual strands, and this “good mutant” might turn fibers into less harmful crystals. Simply introducing normal hemoglobin has been shown not to work, perhaps because the few normal hemoglobin molecules cannot eliminate the fibers.
Ishita Mukerji, of Wesleyan University in Middletown, CT, agrees that this treatment concept could be promising. “It is a really attractive idea,” she says. Although she doesn’t expect gene therapy in the near future, “In the long run, aside from genetic counseling, it’s going to be the best avenue to pursue for people who have sickle cell disease.”
–Lea Winerman
Lea Winerman is a freelance science writer.


