Polymers to Polyhedra
Nanoparticles covered in stringy polymers might someday form the building blocks for drug delivery systems or disease assays. Researchers have made a few versions of these hairballs in various shapes, and now a computer simulation described in the 23 June PRL shows how to controllably make nanoscale 20-sided shapes (icosahedra) and other polyhedra. The trick is to adjust the lengths and abundances of the two types of polymers involved. The report takes aim at a continuing challenge in nanotechnology: how to engineer particles more complex than spheres.
Spherical nanoparticles almost always align themselves like racked billiard balls, which limits the kinds of structures and chemical properties they can attain. With non-spherical shapes, or surfaces that vary in chemical stickiness, researchers could create custom-shaped clusters to serve as catalysts or biological sensors, or selectively connect nanotubes or rods into porous structures for liquid or gas storage. They could also assemble many particles into so-called superlattices to selectively control the transmission of light, for example.
One approach to creating non-uniformly sticky, or multivalent, particles is to align string-like molecules called polymers into a prickly sphere–like a “Koosh ball”–by attaching one end of each molecule to a tiny bead. To imbue these objects with multivalency, researchers attach two kinds of polymer that repel each other. The repulsion forces the two polymer types to segregate into different regions, forming patterns, such as that of a black and white soccer ball.
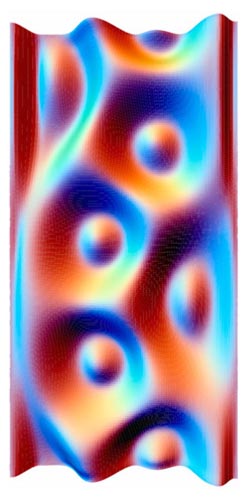
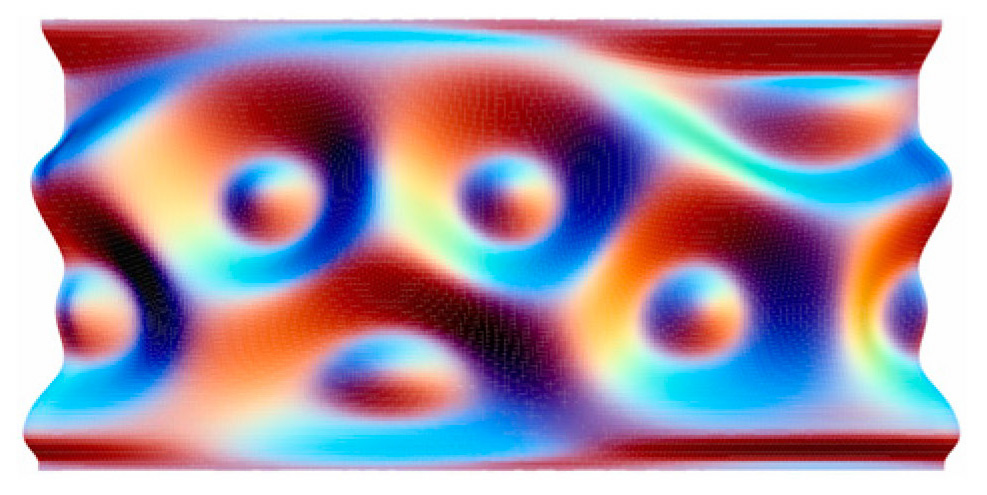
Past experiments have been based largely on trial and error. For a more systematic approach, Jiunn-Ren Roan of the National Chung Hsing University in Taiwan turned to computer simulations. He imagined randomly grafting up to 200 polymers, each 10 to 50 nanometers long, on 10-nanometer-wide nanoparticles, creating something like a spherical mop head with two types of string. “The polymers are densely packed near the surface of the sphere,” says Roan. “So they are like rods near their grafted ends but like noodles near their free ends.” These free ends arrange themselves to maximize contact with their own kind of polymer, but at the same time they don’t like to stretch too much, preferring a kinked conformation as much as possible. The simulation found the balance between these two tendencies.
Roan simulated polymers of different lengths and in different proportions. When the two polymers were equal in length and number, neither could dominate the other, leading to an asymmetric, maze-like pattern around the sphere. But longer or more abundant polymers exerted a greater repulsive force on their counterparts, leading to a wide range of shapes. An abundance ratio of 2 to 1 and a length ratio of 5 to 6, for example, yielded an icosahedron with 12 tall clumps, or “islands,” of the minority polymer protruding above the surface formed by the free ends of the majority polymer. Other forms had three, six, eight, nine, or ten island domains of one polymer.
“A theoretical understanding of such phenomena is very timely and [was] anxiously anticipated,” says Guojun Liu of Queens University in Toronto, whose group has created lumpy, multivalent micelles–prickly spheres entirely of polymer, with no hard center. “The theory should provide guidelines for planning our future work,” he says. “I think it’d be a very interesting system to make,” says Vinothan Manoharan of Harvard University. Similar patterns are seen when oil droplets are coated with liquid crystals or nanoparticles, he says, and a broader theoretical framework encompassing these diverse systems “would be really nice to see.”
–JR Minkel
JR Minkel is a freelance science writer in New York City.