Watching a Molecular Explosion
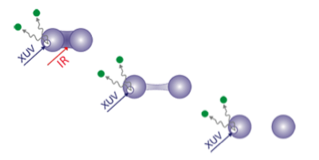
Strong laser fields can destroy the bonds in a molecule, causing it to explode into atomic fragments. Understanding this process in even simple molecules is challenging because it requires experiments that can follow the dissociating molecule on extremely short timescales. In Physical Review A, Maria Krikunova, now at the Berlin Institute of Technology and her colleagues at the University of Hamburg, both in Germany, were able to track the laser-induced explosion of diatomic iodine ( ) using femtosecond pulses of soft x rays produced from a free-electron laser (FEL).
The authors initiated the explosion by shining an intense infrared laser pulse on a gas of the molecules. The laser stripped each molecule of two electrons in its outermost orbitals and broke the iodine-iodine chemical bond. X-ray pulses, whose timing was carefully synchronized with that of the infrared pulse, allowed them to measure how the remaining molecular electrons rearranged as the molecule started to dissociate. They found that after the chemical bond broke, electrons that were initially delocalized over the molecule became localized at the atomic cores. The repulsion between the two charged cores finally resulted in a Coulomb explosion. Within this sequence of events, Krikunova et al. pinpointed a hard-to-observe dissociation regime where the molecular fragments moved apart during the initial laser pulse, facilitating the extraction of additional electrons and leading to enhanced ionization efficiency.
Krikunova et al.’s time-resolved experiment is among the first to be carried out with a new generation of short-wavelength, short-pulse FELs. As they continue to improve FEL-based techniques for capturing timed snapshots of fast processes, researchers are getting closer to the ultimate goal of recording molecular movies. – Matteo Rini