Helium Ions Give Electrons the Shake-Off
The recoil and sudden change in charge of an atomic nucleus emitting a beta particle can literally shake an electron off the atom. Calculations of the probability of shake-off are most accurate when the change in nuclear potential is fast and the electron is in a hydrogenlike orbit, unperturbed by interactions with other electrons, but few reactions involve parent and daughter nuclei with such an ideal structure.
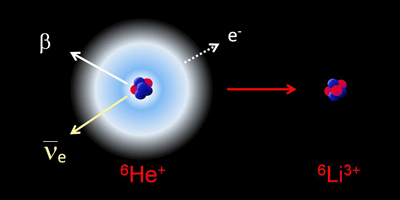
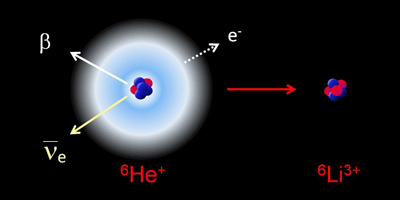
In Physical Review Letters, Claire Couratin of Caen University, France, and colleagues present the first measurement of electron shake-off in the beta decay of ionized helium- ( ). With a half-life of just under a second, beta decays into doubly ionized lithium- ( ), a daughter nucleus in which shake-off can be described with a simple quantum mechanical calculation.
Calculations have predicted that two percent of the daughter lithium would shake off an electron, leaving . Couratin et al. performed a series of experiments in which they collected and confined ions into a trap from a beam of produced at France’s Large Heavy Ion National Accelerator. The trap is outfitted with detectors that measure the coincidence of beta particles and and , allowing the team to measure the probability of shake off in with a relative precision of about a percent. Their result is in excellent agreement with theory, which could make a textbook example for experiments to come. – Jessica Thomas