Cavity-Controlled Chemistry
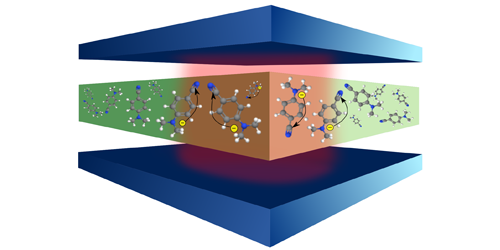
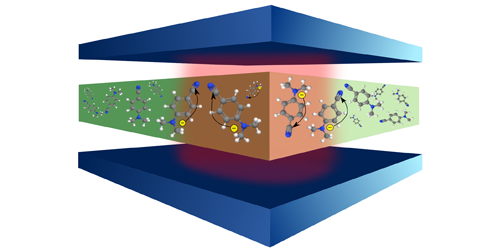
By confining atoms and molecules in microscopic cavities, researchers can stop or accelerate an electronic transition or keep a molecule indefinitely in an excited state. These effects result from the interaction of the molecules with the quantized electromagnetic field in a cavity. Felipe Herrera from the University of Santiago, Chile, and Frank Spano from Temple University in Philadelphia now suggest that similar interactions can be used to control important types of chemical reactions. Specifically, their calculations indicate that electron transfer reactions in a cavity can be much faster than in free space.
Herrera and Spano focused on a type of reaction—found in photosynthesis and cellular respiration—that involves the transfer of electrons between atoms or molecules. The speed of these reactions is often limited by the molecular nuclei, which are prone to reorganize as the electrons travel from the donor to the acceptor groups. This reorganization limits the reaction speed because nuclei move much more slowly than electrons. Through quantum-mechanical calculations, Herrera and Spano showed that the field within the cavity can selectively couple to and accelerate electronic transitions that allow electron transfer to take place before the nuclei can reconfigure. Under such conditions, the researchers projected that the reaction rates might increase by up to 2 orders of magnitude. Their findings could stimulate the development of novel optoelectronic devices, such as cavity-enhanced solar cells and light-emitting diodes.
This research is published in Physical Review Letters.
–Matteo Rini
Matteo Rini is the Deputy Editor of Physics.