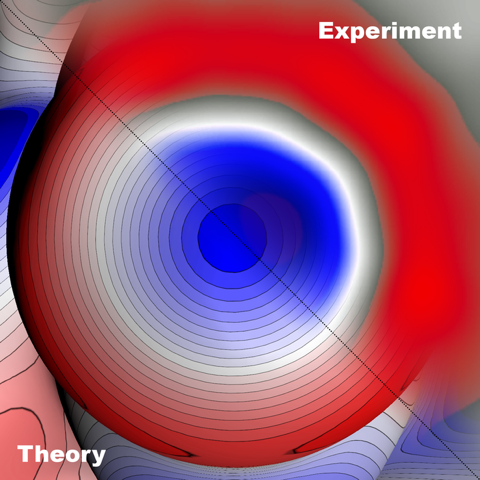
“Black Hole” of Atoms Observed
Like black holes, “sigma-holes” are elusive beasts. Predicted to exist nearly 30 years ago, researchers have struggled to image these ring-like electron-charge patterns. Theory says that the pattern should form around certain halogen atoms when they bond covalently to electronegative atoms because of the repulsive interaction of the atoms. Pavel Jelinek of the Academy of Sciences of the Czech Republic and his colleagues have now ended that struggle, by directly observing the sigma-hole around a bromine atom. They accomplished the feat by updating an imaging technique commonly used to study individual atoms. Knowing an atom’s electron charge distribution is key to understanding interactions between individual atoms and molecules, according to Jelinek. Thus, he says, the achievement could lead to a better understanding of the reactivity of atoms and of why they arrange into particular structures when they combine to form molecules.
To capture their image, Jelinek and his colleagues used a technique known as Kelvin probe force microscopy (KPFM). In this technique, an atomically sharp, vibrating probe is suspended over a thin-film sample. The probe and sample are electronically connected such that they form a capacitor. The probe is then lowered toward the sample surface, where it records shifts in the frequency of the surface’s vibrations over a range of voltages.
The peak in the vibration-frequency shifts of the probe relates to what’s known as a material’s work function, a quantity that contains information about the composition and electronic state of its atoms. Researchers have used KPFM to reconstruct the composition of a surface with atomic resolution, for example. But to capture the distribution of electrons around an atom requires imaging much smaller features.
To achieve subatomic resolution with KPFM, Jelinek and his colleagues first refined their theoretical understanding of the technique and then used that understanding to update their KPFM setup. For example, the team disentangled all the different forces acting on the tip and then chose the tip’s terminal atom so that it would be sensitive to the electrostatic forces that are used in KPFM. The choice of terminal atom also ensured that no electrostatic features might blur the structure that they wanted to image. Previously, researchers used carbon monoxide molecules for KPFM imaging, but Jelinek’s team found that carbon monoxide’s quadrupolar-shaped polarization is so similar to the shape of the sigma-hole that it masks the target’s detection. In contrast, xenon atoms have a homogeneous polarized charge. “Immediately from theory we saw that [xenon] was the best tip candidate; much better than the previously used carbon monoxide,” Jelinek says.
The team tested their updated setup to image two molecules—one containing bromine and one fluorine—that they placed on a silver surface. Theory predicts that bromine should have a sigma-hole in its electron charge distribution because it interacts repulsively with its neighboring atom, while fluorine doesn’t because its strong attraction to its neighbor suppresses the formation of a sigma-hole. The team’s experimental images match those predictions. “The images are a surprisingly good match for the theory,” Jelinek jokes.
Now that the team know their technique has the sensitivity needed to image the electron charge distribution around an atom, they would like to use it to study other properties of atoms. One property Jelinek has his eye on is local polarizability—how the electronic cloud around an atom locally responds to an applied electric field, for example. “There are currently no techniques that can measure that at the atomic level,” he says. “Doing that is, I think, our next step.”
–Katherine Wright
Katherine Wright is the Deputy Editor of Physics Magazine.