New View of Magnetic Atoms
Researchers have taken one of the clearest looks at the magnetic behavior of atom-thin layers of metal. The team grew very uniform layers of cobalt and measured the direction of the magnetism inside. They found that as the thickness of the metal film increases stepwise from one to three atomic layers, its magnetism changes direction twice, by 90 degrees each time. The team’s calculations completely explain this surprising, rarely observed effect, as they report in the 14 April PRL. Understanding atomic-scale magnetic interactions like these is essential for researchers hoping to decrease the size of magnetic memories.
Computer hard drives encode 0s and 1s as the alignment directions of tiny bar magnets. Each bar magnet is a chunk of metal whose atomic spins are aligned; the amount and direction of magnetism from those spins is called the magnetization of that chunk. To build smaller units of memory, researchers need to understand how smaller numbers of spins orient themselves, such as in sheets one or a few atoms thick. Complicating this task, metal atoms deposited across a surface normally form many small hills and valleys, with no broad plateaus of uniform height. Determining a sample’s thickness or magnetization often involves some guesswork, says Juan de la Figuera of the Autonomous University of Madrid.
To make an unambiguous measurement, de la Figuera, his student Farid El Gabaly and colleagues used an electron microscope to image a layer of cobalt atoms growing on a ruthenium surface. Using the images, they continually fine-tuned the cobalt source during the growth process to create uniform layers one, two, or three atoms thick and 10 microns across.
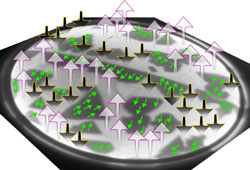
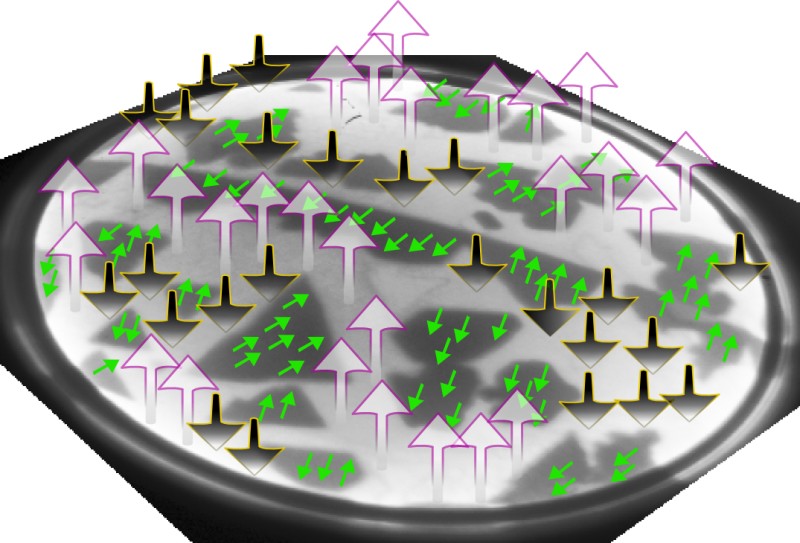
With wide, atomically flat areas, the team could measure magnetization knowing exactly what thickness they were observing. They used a so-called spin-polarized electron beam, in which the electron spins point the same direction, and imaged the surface with the beam polarized both perpendicular to the surface and parallel to it. Combining the images, the team generated a map of the magnetization in different regions of the surface, like a topographical map of a country. The one- and three-atom-thick layers were both magnetized parallel to the surface, like a compass needle, while the two-atom layer was magnetized perpendicular to the surface. “The surprising thing was why for a very, very thin film it would want to be in plane and then it would go out of plane,” says de la Figuera. Such flip-flopping magnetization also occurs in iron, he says, where its precise cause has remained obscure.
Knowing the structure of the cobalt layers unambiguously was key to explaining the effect theoretically, says de la Figuera. Left to themselves, the cobalt atoms would magnetically orient “in-plane,” like an array of bar magnets lying close together, flat on the floor. But other influences, such as the distorted cobalt crystal structure caused by the ruthenium below, can favor an out-of-plane alignment. That’s because the electrons involved in the magnetization also participate in the bonds between atoms, so the magnetic alignment is influenced by the positions of neighboring atoms. The team calculated that for exactly two atomic layers, these other influences overcome the atoms’ intrinsic tendency to align in-plane.
“The paper confirms in a beautiful and spectacular way our understanding of nanomagnetism,” says André Thiaville of the University of Paris-South. “It is very nice to have real space observations on very clean samples, in order to see the very fundamental mechanisms at work.”
–JR Minkel
JR Minkel is a freelance science writer in New York City.