What Makes It So Hard?
A recently developed material is hard enough to scratch diamond but is much easier to make. It’s also half as hard along some directions in the crystal as in others. A theory in the Rapid Communications section of the August Physical Review B explains this unusual property and suggests that the hardness depends on the strength of bonds perpendicular to the direction the material is squeezed, not the parallel bonds. The simple model could help researchers find other superhard materials.
The stiffness of a material describes how much it resists being temporarily stretched or compressed, springing back when force is released. In contrast, its hardness is its resistance to permanent shape change, which happens when atoms are pushed to new locations. The intrinsic hardness of a single crystal can be measured by pressing against its surface with the point of a microscopic diamond pyramid until it stops and measuring the area of the dent. In recent years, several teams have related this measure of hardness to the materials’ atomic-level structure.
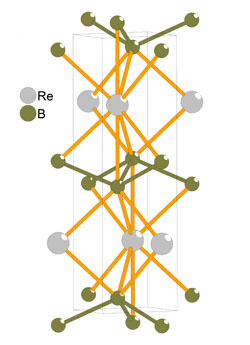
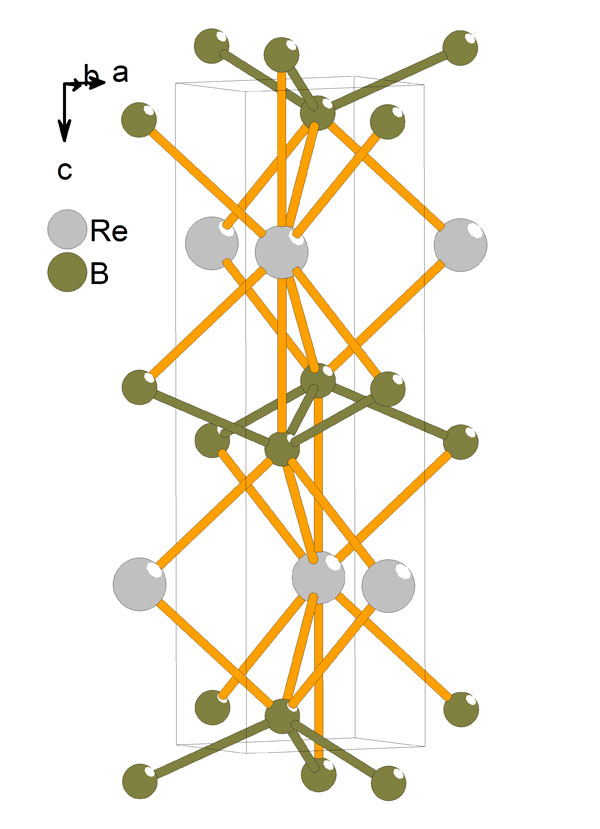
Since 2005, other researchers have been developing layered “transition-metal diborides,” such as materials whose hardness rivals that of diamond but that don’t require high temperatures and pressures to produce. One striking property of these materials is their extremely direction-dependent hardness, and there is no theory to explain it. A theory that captures this behavior should also be more reliable for predicting hardness in other materials.
Antonín Šimůnek of the Academy of Sciences of the Czech Republic decided to fill this need by extending his previous work on more conventional materials. He previously developed a formula that combined the contributions of the various types of bonds in a crystal to calculate its hardness, but included bonds in all directions in the crystal with equal weight.
To model the directional differences, Šimůnek was inspired by experiments on sheets of graphene, the single-atom-thick version of graphite. Pressing down with a sharp point in the middle of a graphene sheet is like standing on a trampoline–the material’s strength depends entirely on the bonds perpendicular to the direction of pressure. After all, there aren’t any bonds in the parallel direction for such a thin sheet.
But Šimůnek realized that even in a thick material, “the sideways direction is important because these sideways bonds are broken first,” he says. “This is surprising, because people usually think that hardness is related to bonds that prevent the close approach of atoms during compression.” These compressed bonds determine the stiffness, he says, but compression doesn’t easily break bonds. Instead, breaking of the stretched, perpendicular bonds is what ultimately limits the hardness.
Šimůnek adapted his earlier model by mathematically weighting the contributions of each bond to the total hardness based on its direction, with bonds perpendicular to the applied force being given the most weight. In the transition-metal diborides, his model explains the greater hardness in the direction perpendicular to the layers that contain strong boron-boron bonds. As for other materials, Šimůnek says, his “extremely simple” calculation is useful for guessing the properties of new materials that haven’t been made yet.
“What’s nice about this paper is that it’s doing calculations that can look at the hardness in different directions,” adds Richard Kaner of the University of California, Los Angeles, co-inventor of the transition-metal diborides. “It’s not that easy to measure hardness in different directions.” He is hopeful that other theorists may be inspired to analyze this aspect of hardness.
–Don Monroe
Don Monroe is a freelance science writer in Murray Hill, New Jersey.