Atomic Dance Drives Melting
Melting is probably the most familiar phase transition, but researchers still don’t agree on exactly how an idealized, perfect solid becomes liquid. In the April Physical Review B, researchers describe the atomic choreography that accompanies this process in a computer version of a crystal. They attribute melting to increasingly complex motions of groups of atoms acting in concert, rather than to vibrations of individual atoms or to particular atomic arrangements invoked by previous researchers.
When you heat a crystal, it ordinarily melts when the combination of entropy and energy favors the liquid phase. But the liquid can’t take over until a large-enough droplet adopts a liquid-like arrangement. In the real world, melting usually starts at crystal boundaries or imperfections, where atoms can rearrange more easily. But physicists have speculated for decades about how a perfect, infinite crystal melts. Such “homogenous” melting may resemble what happens in rapid heating during laser processing of materials or explosions.
In computer simulations, researchers can make a crystal effectively infinite by mathematically connecting its opposite faces, so that an atom leaving at the left edge reappears at the right edge. The problem is that even the fastest computers can track a large batch of atoms for the equivalent of only a few nanoseconds, not long enough for the complex rearrangements that may be important in homogeneous melting. To speed things up, many researchers heat rapidly, much faster than is possible in experiments, and far past the transition temperature, until their virtual solid melts suddenly. But the catastrophic melting at these high temperatures may not resemble what happens with more gentle, though still fairly rapid heating, say Mo Li and his former graduate student, Xian-Ming Bai, of the Georgia Institute of Technology in Atlanta.
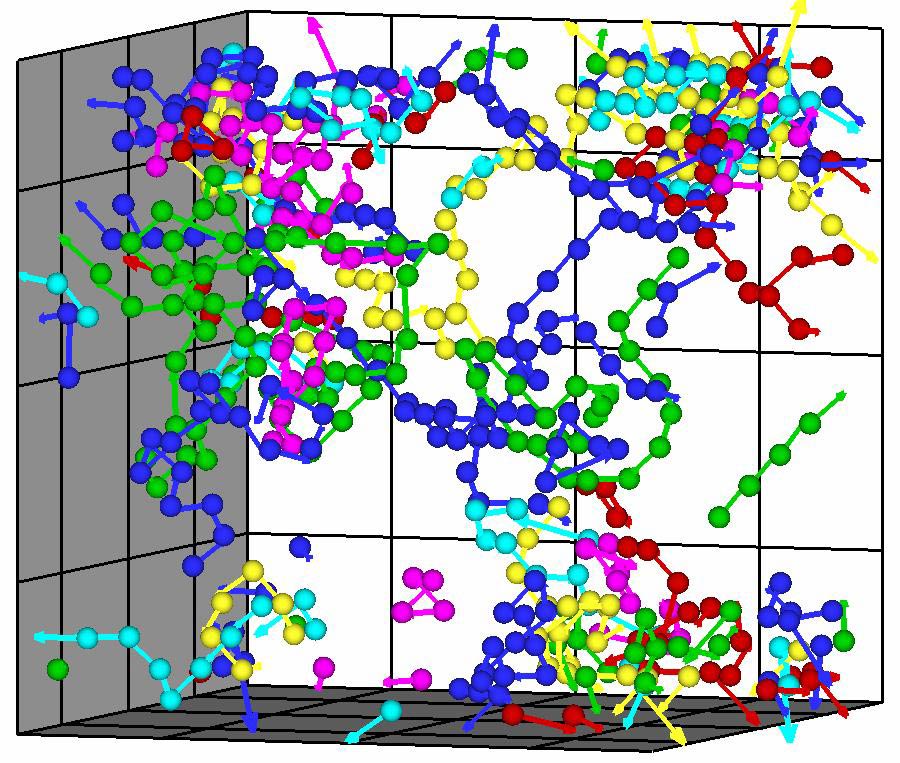
To study a less extreme kind of melting, Bai and Li started with a virtual crystal comprising thousands of weakly-attracting spheres meant to resemble argon atoms. They quickly heated it somewhat above the transition temperature, and then waited. Under these conditions, they found, melting is always preceded by extensive movement of atoms away from their starting points. Rather than moving individually, however, the atoms form long, zigzagging chains that move as a group. The first atom finds a neighboring lattice site that is perhaps briefly open and hops into it, and then a “bucket brigade” follows, with each atom occupying the space vacated by its neighbor. Over time, the chains proliferate and get entangled in knots, Li says. “These knots form the nucleus of the liquid droplet.”
The researchers tested many previous explanations for melting but found them wanting, at least for their heating procedure. One well-known model attributes melting to large-amplitude oscillations of atoms that remain at their sites. Another suggests that the liquid forms because the crystal becomes mechanically floppy. Other explanations invoke growing numbers of crystalline defects, such as “interstitial” atoms that move to locations between lattice sites. The team saw no evidence for any of these.
”I’m impressed by the accuracy” of the new simulations, says Göran Grimvall of the Swedish Royal Institute of Technology in Stockholm. Although his own simulations using gradual heating had traced melting to interstitial atoms [1] he notes that crystals may melt in different ways depending on details such as the precise forces between atoms used in the simulation. Grimvall says the real challenge in melting research is knowing what structures to look for in the complex simulation results. “I think this will continue for quite some time.”
–Don Monroe
Don Monroe is a freelance science writer in Murray Hill, New Jersey.
References
- M. Forsblom and G. Grimvall, “Homogeneous melting of superheated crystals: Molecular dynamics simulations,” Phys. Rev. 72, 054107 (2005)