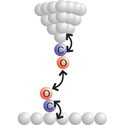
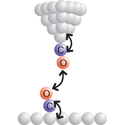
Sharp end
There are several tricks of the trade in atomic force microscopy (AFM) for obtaining images of surfaces with atomic level resolution. Scientists recently added to this toolkit when they showed that terminating an AFM tip in a single carbon monoxide molecule allowed them to image individual atoms in pentacene.
Writing in Physical Review Letters, Zhixiang Sun and colleagues at the University of Utrecht, Netherlands, apply this relatively new technique to map out, in three dimensions, the chemical forces between two carbon monoxide molecules.
As the oscillating tip of an AFM approaches the atoms or molecules on a surface, it experiences both attractive (van der Waals) and repulsive (Pauli) forces. Measuring these forces with sufficient accuracy—one of many applications of AFM—requires that the tip be sufficiently near the surface that these forces exert a sizable shift on its resonance frequency, but not so close that the tip actually bends or moves the molecules.
In their work, Sun et al. identify the optimal distance range within which to measure the chemical forces between a carbon monoxide molecule dangling from the AFM tip and an identical molecule on a copper surface. Their force map is qualitatively similar to that expected between two isolated molecules, free of the complications arising from the presence of the AFM tip and the substrate. Sun et al. note, however, that this result may not hold in all experiments, as short-range repulsive interactions can cause either molecule to bend and obscure the interpretation of the force measurements. – Jessica Thomas