Squeezed Environment Boosts Engine Performance
The theory of thermodynamics has taught us how to harness the abundant and disordered energy that exists all around us as heat and how to convert it to useful energy in the form of work. For example, we can burn fuel to make the wheels of our cars turn. This understanding helped build the machines of the Industrial Revolution, which forever changed our societies and place in the world. Another revolution is occurring at the nanoscale, where researchers are busy making nanomachines for a host of applications, such as targeted drug delivery and green-energy storage.
But how efficiently can we extract energy from a hot environment? The typical tool we use is a heat engine, which allows heat to flow from a hot bath to a colder one, extracting work in the process. In 1824, Carnot showed that the maximum efficiency of a heat engine is given by the ratio of the temperature of the two baths [1]. Jan Klaers and colleagues [2] at the Swiss Federal Institute of Technology (ETH) in Zurich have, for the first time, experimentally demonstrated that, by engineering the properties of the environment, they can drive a nanoscale engine twice as efficiently as Carnot predicted. This will allow synthetic nanoengines to operate more efficiently than their macroscopic counterparts.
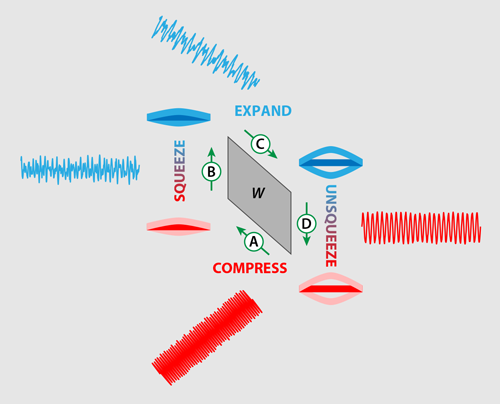
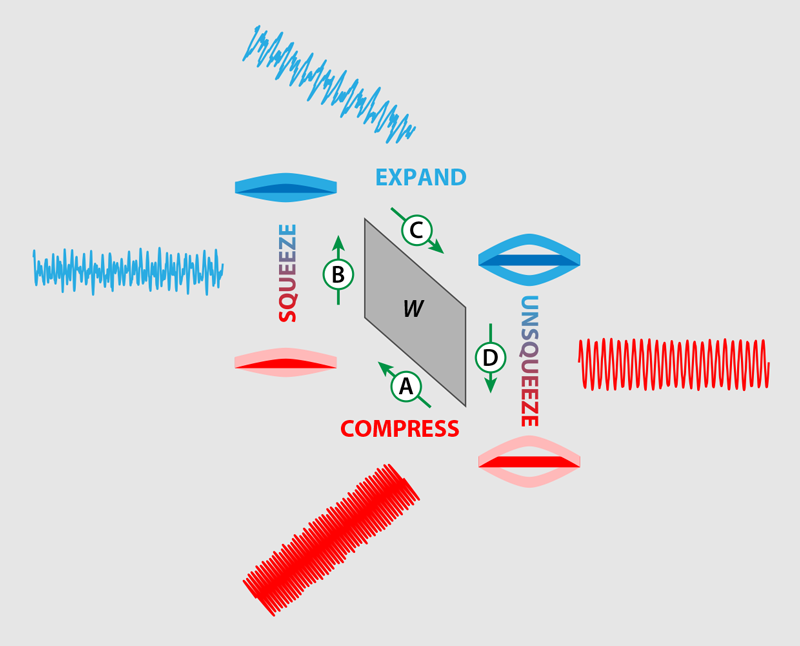
Klaers and colleagues’ engine is formed by a nanomechanical beam that is roughly 20 𝜇m long and fixed at both ends (Fig. 1). The team sets the beam in vibrational motion by applying an electronic signal that exerts a force on the beam. They then add noise to the signal, making the position and momentum of the beam fluctuate. The noisy signal acts as a thermal environment for the nanobeam, with the amount of noise added setting its temperature.
Next, the team borrows a trick from the quantum optics community—they “squeeze” the electronic signal. Squeezing is a process by which fluctuations can be reduced in one of a system’s degrees of freedom at the expense of another. For example, a tiny pendulum immersed in a thermal environment has a fixed relationship between its position and momentum. But it is possible to “squeeze” the pendulum’s motion such that fluctuations in its position are reduced at the expense of increasing fluctuations in its momentum [3].
By driving the nanobeam with a squeezed electronic signal, Klaers and co-workers are able to extract 244 meV of heat from the noisy “bath” and convert it to 26 meV of work—an efficiency of around 10%, which is twice the Carnot limit for this engine. The process by which they do this involves introducing different levels of noise to change the effective temperature, different signal frequencies to effectively compress or expand the beam, and different levels of squeezing to effectively change the pressure (Fig. 1). It is worth noting that no laws of thermodynamics are being violated in this system because the squeezing is an inherently nonequilibrium process, outside of the remit of standard thermodynamic theory. In the future, researchers may not have to go to such lengths to engineer nonequilibrium environments since nature provides them for free, particularly in a biological setting. Many vital processes within cells are powered by molecular nanomachines that are out of equilibrium with their environments.
In this experiment, the nanobeam does work in the form of strain on its clamped end points—not a particularly useful task. In principle, however, one could couple these end points to other oscillating systems that can’t be electrically controlled and drive their motion with an efficiency in excess of Carnot’s prediction. This “drive” capability is an improvement over previous nanoengines, which have been formed of single levitated particles [4, 5]. Such particle systems have proven useful for testing fundamental predictions of thermodynamics, but they are difficult to connect to other systems. They are a bit like car engines without wheel connections. By contrast, the nanofabricated structure presented in this work is inherently integrable, heralding genuine applications in more complex nanomachines. The 26 meV of work that Klaers and co-workers’ nanoengine generates may sound tiny, but this amount of work is not negligible on the nanoscale; it would be enough to propel a nanoparticle or biological cell at a velocity of a few centimetres per second.
As technology ever miniaturizes, dealing with environmental heat becomes an increasing problem. The energy imparted to a nanoscale device due to thermal fluctuations of the environment becomes comparable to the operating energy of the device itself, which can hamper its operation. At the nanoscale, we enter a regime where we cannot just rely on the standard theorems of thermodynamics, or averaged quantities, to understand the generation of heat and work and the behavior of machines. A large machine doesn’t care if some air molecules hit it at twice the average velocity, but such a fluctuation has a big effect on a nanoscale device. Klaers and colleagues’ research highlights the potential of harnessing these otherwise disruptive fluctuations to maximize energy conversion.
Heat is an even greater problem for quantum systems because thermal energy from the environment leads to the destruction of their fragile quantum states. Playing the same kind of thermodynamic tricks that Klaers and colleagues used, but in the quantum arena [6, 7], could lead to the development of quantum machines [8] that exploit effects such as entanglement in their operation. This isn’t as far off as one may think; the motion of oscillating nanobeams similar to that used here has been cooled to the quantum level [9]. Klaers and colleagues’ research pushes the hundreds-of-years-old theory of thermodynamics to its limits and beyond, and we may hope that nanoscale engines will one day enable a Quantum Industrial Revolution. And who would dare predict what changes that will bring?
This research is published in Physical Review X.
References
- S. Carnot, Reflections on the Motive Power of Heat, edited by H. Thurston (J. Wiley & Sons, New York, 1890).
- J. Klaers, S. Faelt, A. Imamoglu, and E. Togan, “Squeezed Thermal Reservoirs as a Resource for a Nanomechanical Engine Beyond the Carnot Limit,” Phys. Rev. X 7, 031044 (2017).
- M. Rashid, T. Tufarelli, J. Bateman, J. Vovrosh, D. Hempston, M. S. Kim, and H. Ulbricht, “Experimental Realization of a Thermal Squeezed State of Levitated Optomechanics,” Phys. Rev. Lett. 117, 273601 (2016).
- V. Blickle and C. Bechinger, “Realization of a Micrometre-Sized Stochastic Heat Engine,” Nature Phys. 8, 143 (2011).
- J. Rossnagel, S. T. Dawkins, K. N. Tolazzi, O. Abah, E. Lutz, F. Schmidt-Kaler, and K. Singer, “A Single-Atom Heat Engine,” Science 352, 325 (2016).
- S. Vinjanampathy and J. Anders, “Quantum Thermodynamics,” Contemp. Phys. 57, 545 (2016).
- J. Millen and A. Xuereb, “Perspective: Quantum Thermodynamics,” New J. Phys. 18, 011002 (2016).
- J. Millen and A. Xuereb, “Rise of the Quantum Machines,” Phys. World 29, 23 (2016).
- A. D. O’Connell et al., “Quantum Ground State and Single-Phonon Control of a Mechanical Resonator,” Nature 464, 697 (2010).