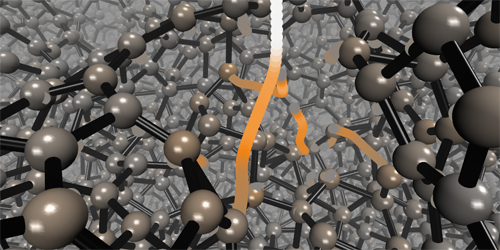
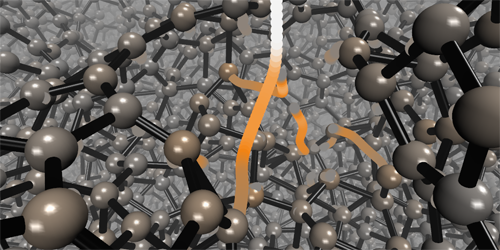
How Diamond-Like Carbon Films Grow
Very dense films of amorphous carbon can have properties—super hardness, chemical inertness, and optical transparency—similar to those of diamond, one of carbon’s crystalline forms. Researchers have learned empirically how to grow films of this so-called diamond-like amorphous carbon (DLC) by depositing carbon atoms onto a carbon substrate. However, they don’t understand theoretically how these films form. Now, combining a machine-learning algorithm with molecular dynamics simulations, Miguel Caro from Aalto University in Finland and colleagues have shown that DLC films form via a “peening” mechanism. In such a mechanism, the impact of a carbon atom on a surface induces pressure waves that locally change the film’s density and, in turn, the orbital hybridization of the atoms in the film.
The team simulated the sequential deposition of individual carbon atoms onto a carbon substrate. Using a previously developed machine-learning algorithm, they calculated the interatomic potentials between the atoms and fed that information into the molecular dynamics simulations. This procedure allowed them to map the mass density of the films after each impact. They found that the incoming atoms created a pressure wave that pushed atoms away from the impact site, such that each impact decreased the density of atoms at the film’s surface and increased it in the film’s interior. In the film’s interior, the density change caused a high fraction of carbon atoms to acquire a tetrahedral sp3 hybridization state—the same state that carbon atoms have in diamond. The simulated density profiles agree with those derived from previous experiments.
These results could help researchers optimize the growth of DLC films, which are attractive for building implantable electronic devices, since DLC is biocompatible.
This research is published in Physical Review Letters.
–Katherine Wright
Katherine Wright is a Contributing Editor for Physics.