Nuclear Spectroscopy Reveals New Shapes of Excited Nuclei
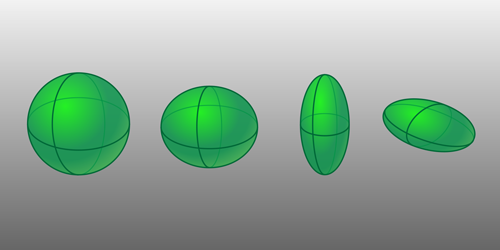
Atomic nuclei take on excited states when they vibrate, rotate, or when their constituent nucleons exchange one nuclear shell for another. In nuclei with nearly filled nuclear shells, it has long been thought that low-energy excitations were due exclusively to different patterns of vibration around a spherical shape: only in rare, high-energy excitations were these nuclei expected to assume more exotic shapes. Now, Paul Garrett, of the University of Guelph in Canada, and colleagues have found that the lowest-energy excited states of cadmium-110 and cadmium-112—once considered textbook examples of spherical vibration—are instead due to the rotation of various nonspherical shapes. The result is also the best evidence to date that a stable nucleus like cadmium can assume multiple shapes—all previously studied nuclei with coexisting shapes have been radioactive.
To explore the shapes of cadmium nuclei, the team started by depositing beams of radioactive indium and silver isotopes onto a target surrounded by radiation detectors. The indium and silver decayed into isotopes of cadmium, with some of these daughter nuclei existing briefly in excited states. As the nuclei settled into lower energy states, they emitted gamma rays that revealed details about the excited states, such as their energies, their occurrence rates, and their decay rates. A comparison of these data with state-of-the-art calculations suggested that the excitations were best explained not by spherical vibrations but by rotating ellipsoidal nuclei. The model predicts that these nonspherical nuclear configurations exist right down to the ground state, with four distinct ellipsoidal shapes representing the lowest energy states. Proving this empirically is the team’s long-term goal but doing so will take many experiments across many different laboratories.
This research is published in Physical Review Letters.
–Marric Stephens
Marric Stephens is a freelance science writer based in Bristol, UK.