Scientists Unravel Silk Habitat Morphology
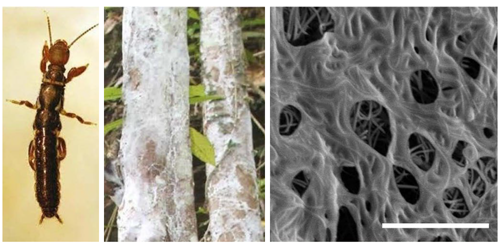
Spiders and silkworms may be nature’s best-known silk spinners, but the prize for the finest handiwork goes to a lesser-known order called Embioptera. These insects, also known as webspinners, form their fine silk threads (30–140 nm in diameter) into webs, which transform into cloth-like sheets or films when wet. Because webspinner threads are thinner than optical wavelengths, the mechanism driving this transformation has been elusive. Now, using optical microscopy and scanning electron microscopy, Richard Barber of Santa Clara University, California, and colleagues show that water-soluble proteins in the silk are responsible for this mechanism [1]. The mechanism could one day be repurposed for biomedical applications like drug delivery.
In the wild, webspinners use their silk sheets to build homes in gaps in tree bark. To replicate these structures in the lab, Barber and his colleagues encouraged webspinners from two different species to build across grooves in a graphite substrate. After collecting the silk samples, the researchers observed how the material reacted to microliter drops of water. They found that, instead of soaking through and dissolving the silk threads entirely, the water dissolved only the water-soluble protein cores, leaving the lipid-based outer sheaths of the threads intact. These dissolved core proteins formed a layer on the surface of the water droplets. Then when the water had evaporated, the residue remained as a thin film on the silk structure. The purpose of these films is unknown, but biologists believe that they might protect the insects from rainfall in tropical climates.
Next, the researchers plan to study other species of webspinners to determine how common this waterproofing mechanism is. They also hope to use spectroscopy to study the molecular structure of the silk’s proteins.
–Sarah Wells
Sarah Wells is an independent science journalist based outside of Washington, DC.
References
- A. C. Andrews et al., “Morphological transformation from fibers to sheets in embiopteran silk,” Phys. Rev. E 106, 014801 (2022).