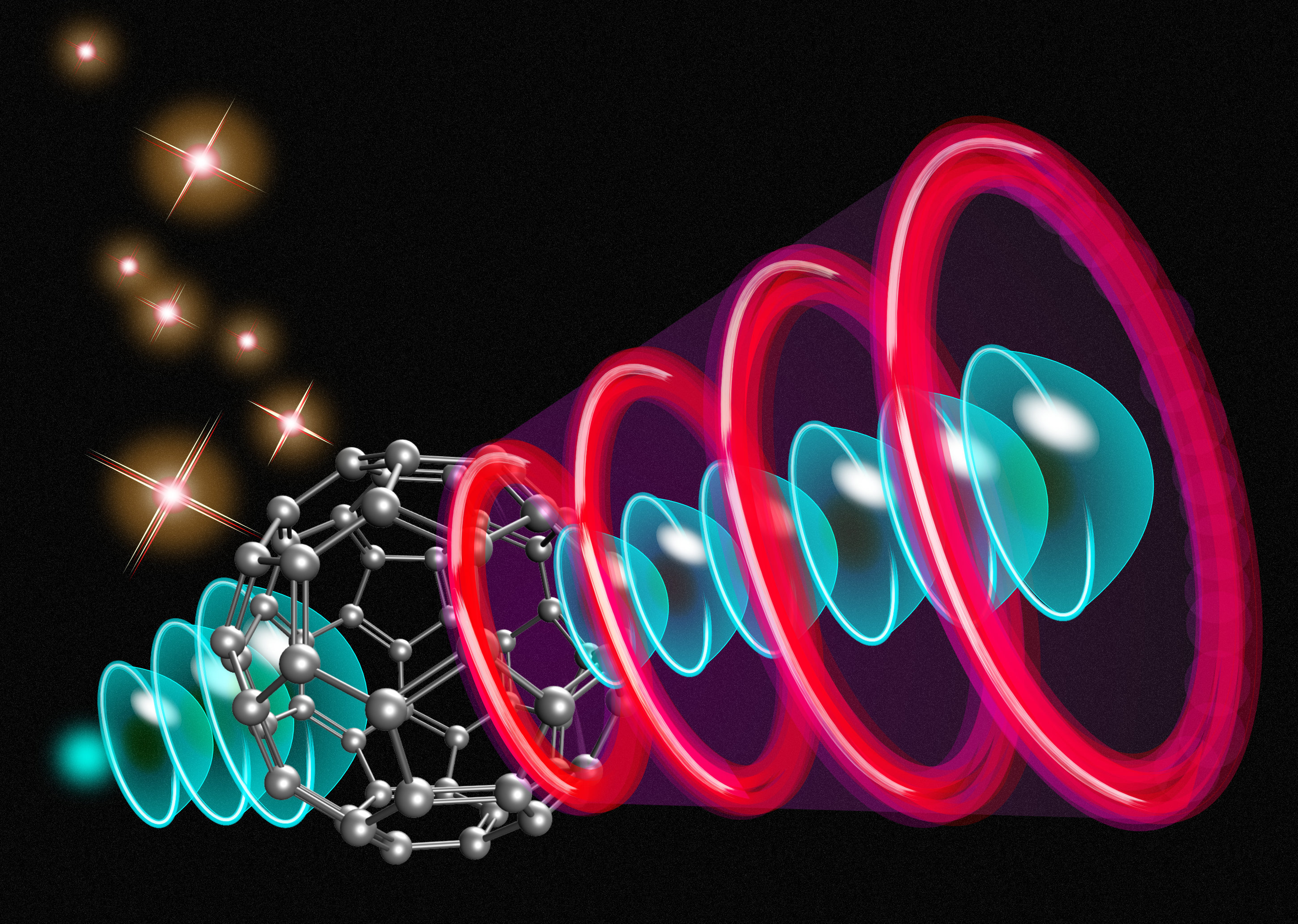
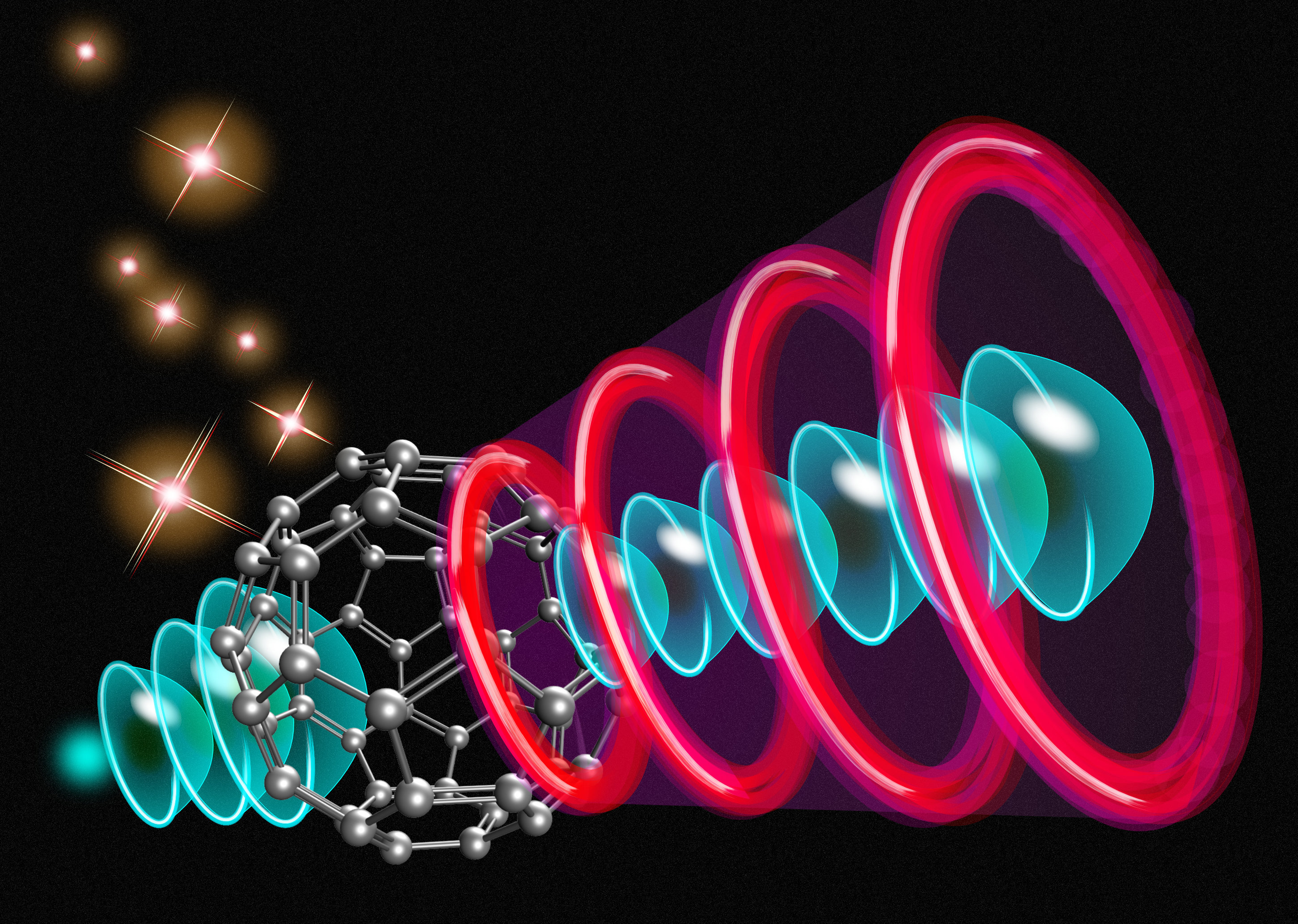
Molecular-Orbital Electron Sources
The ultimate miniature electronic device may be one that manipulates individual electrons with subnanometer and subfemtosecond precision. The past few decades have seen immense progress in the control of ultrafast electronic processes, including in the context of vacuum nanoelectronics, where electrons travel from a nanoscale emitter to a target electrode through a vacuum. Now Hirofumi Yanagisawa at the Japan Science and Technology Agency and colleagues have taken an important step toward optimal spatial control by using the orbitals of a single molecule to shape its electron emission (Fig. 1) [1]. The approach offers the prospect of building highly controllable electron emitters, but also of furthering our understanding of the role of molecular orbitals in the electronic structure of solids.
Fundamental to achieving extreme control over electron emission is defining the spot from which electrons are ejected from the emitter. One approach is to physically shape the material of the emitter into the desired spot pattern. Doing that at the subnanometer scale would entail significant material- and fabrication-related challenges, however. Instead, Yanagisawa and colleagues have demonstrated the clever idea of using the inherent electronic structure of a molecule to route the electrons for emission. In essence, the molecular orbitals are used as a spatial filter to control the emission pattern.
The team’s work grows out of two broad areas of investigation that have progressed significantly over the past few decades. One of these involves the study of femto- and attosecond electron dynamics and the creation of ultrafast electron sources, exemplified by the 2006 demonstration of tight spatial control over femtosecond electron pulses through emission from a nanoscale metallic tip [2–8]. The second is the study of electron emission patterns originating from molecular structures and nanostructures. Examples include patterns corresponding to the tip structures of nanotubes and nanowires, which change as the tip evolves during nanotube growth [9–11]. It is by combining the techniques of ultrafast emission and emission microscopy that Yanagisawa and colleagues have demonstrated that the emission patterns can be directly linked to specific molecular orbitals.
The researchers created a layer of C60 molecules (“buckyballs”) on a tungsten tip. Under a strong electric field, individual C60 molecules protruded from the layer and emitted electrons, which originated in the underlying tungsten reservoir. Projected onto a phosphor screen, the emission pattern showed structures—for example, a spot, a ring, a cross, or a “two-leaf” pattern—closely reflecting the spatial symmetries of the unoccupied molecular orbitals of C60, which the electrons passed through on their route to emission. The researchers reproduced these patterns based on density-functional-theory (DFT) calculations.
After observing the emission patterns under a large electric field, Yanagisawa and colleagues then reduced the field strength slightly to avoid field-induced emission, and instead induced two-photon photoelectron emission by illuminating the tip with laser pulses. Whereas under field-induced emission the electrons passed through an unoccupied C60 orbital close to the Fermi level of the tungsten reservoir, under photoelectron emission the electrons were boosted above the Fermi level by the two-photon excitation energy. As a result of this boost, the electrons passed through a different, higher-energy unoccupied molecular orbital. By switching between field emission and photoemission, the researchers found that the emission pattern switched back and forth correspondingly. These results thus present compelling evidence that the electrons are routed through specific molecular orbitals of the C60 molecule, as dictated by the excitation energy, before being emitted. Consistent with this result, they additionally showed that even spontaneous changes of the emission pattern during pure field emission are attributable to different molecular orbitals becoming available as emission pathways due to small displacements of the molecule caused by thermal energy.
This demonstration of subnanometer spatial control of electron emission using the inherent electronic structure of a molecule opens an exciting avenue for engineering electron sources with well-defined and desirable emission properties. Having helped establish the electronics industry in the first half of the 20th century, vacuum devices are still critical in high-speed and high-power applications because electrons travel unhindered in a vacuum. As miniaturization of solid-state technologies becomes more challenging, nanoscale vacuum electronic devices become more appealing. It will be interesting to see how the present work will be developed. Will routing through specific molecular orbitals have implications for the timing of emission and for the noise in the emission current? What will the consequences be for electron-optical properties? Will other molecular structures—carbon nanotubes naturally come to mind—produce similar results? Application possibilities are vast, ranging from vacuum transistors to electron-emitter arrays with high coherence to tabletop accelerators [12–14] to name only a few.
Regardless of these possible future applications, the work is also exciting from a fundamental point of view. We know that electrons interact strongly with each other. The foundation for proper description of a system—be it a molecule, a nanoscale object, or macroscopic bulk—is the many-body wave function. In DFT, molecular orbitals, which are one-electron functions, provide mathematical convenience and ease of interpretation, but their validity is approximate. That DFT and even basic one-electron models have proven so successful in describing many material systems is fortunate, but is not to be taken for granted—for example, strongly correlated materials sometimes demand more elaborate treatments or at least corrections to one-electron model results. The work of Yanagisawa and colleagues gives us a direct visual access to DFT orbitals in a molecule, adding further empirical legitimacy to the concept of one-electron wave functions—and that’s a remarkable result.
References
- H. Yanagisawa et al., “Light-induced subnanometric modulation of a single-molecule electron source,” Phys. Rev. Lett. 130, 106204 (2023).
- P. Hommelhoff et al., “Field emission tip as a nanometer source of free electron femtosecond pulses,” Phys. Rev. Lett. 96, 077401 (2006).
- R. Pazourek et al., “Attosecond chronoscopy of photoemission,” Rev. Mod. Phys. 87, 765 (2015).
- G. G. Brown et al., “Characterizing multielectron dynamics during recollision,” (2020) arXiv:2010.06165.
- Y. Dai et al., “Ultrafast nanofemto photoemission electron microscopy of vectorial plasmonic fields,” MRS Bull. 46, 738 (2021).
- H. Y. Kim et al., “Attosecond field emission,” Nature 613, 662 (2023).
- S. Meier et al., “Few-electron correlations after ultrafast photoemission from nanometric needle tips,” (2022) arXiv:2209.11806.
- F. J. García de Abajo and C. Ropers, “Spatiotemporal electron-beam focusing through parallel interactions with shaped optical fields,” (2023) arXiv:2302.05969.
- K. Hata et al., “Field emission microscopy of adsorption and desorption of residual gas molecules on a carbon nanotube tip,” Surf. Sci. 490, 296 (2001).
- Y. Sun et al., “Polymethyl methacrylate thin-film-based field emission microscope,” IEEE Trans. Nanotechnol. 11, 441 (2012).
- M. Marchand et al., “Growing a carbon nanotube atom by atom: “And yet it does turn”,” Nano Lett. 9, 2961 (2009).
- R. Bhattacharya et al., “Complementary vacuum field emission transistor,” IEEE Trans. Electron Devices 68, 5244 (2021).
- S. Tsujino et al., “Measurement of transverse emittance and coherence of double-gate field emitter array cathodes,” Nat. Commun. 7, 13976 (2016).
- R. J. England et al., Progress in developing an accelerator on a chip, Proc. 13th Int. Particle Acc. Conf., 16, (2022).