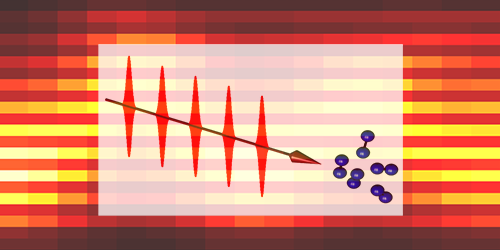
Quantum Rocking Motion in Molecular Rotors
Physicists use lasers to ratchet up the rotation of molecules. A new study now shows that this acceleration can be reversed by quantum effects to produce forward-backward motion. This is similar to so-called Bloch oscillations that occur for accelerated electrons in a crystal. The observation of this effect in a room-temperature gas introduces a simple technique for studying quantum transport phenomena.
The prediction of Bloch oscillations dates back to 1929. When a crystal is placed in a static electric field, the crystal’s free electrons accelerate. However, at a certain peak momentum (determined by the spacing of the atoms in the lattice), the electrons will scatter backwards. The repetition of the forward acceleration and backward reflection results in an oscillation cycle. The effect was first observed in 1992 in semiconductors, and a lot of subsequent research has focused on ultracold atoms. However, these systems can be challenging to study.
A team headed by Philip Bucksbaum from Stanford University in California and Ilya Averbukh from the Weizmann Institute in Israel looked for Bloch-like oscillations in a simpler system—room-temperature nitrogen molecules. They exposed a nitrogen gas sample to a periodic train of femtosecond laser pulses. Each pulse gave a little kick to the rotation, causing the molecules to hop from one angular momentum state to the next higher one, as if they were moving through a lattice in momentum space. At a certain point, the molecular wave packet reflected backwards (similar to the electron-crystal case), and the angular momentum began to decrease. To measure these oscillations, the team used a probe beam whose polarization shifted depending on how fast the molecules were rotating. The results agreed with the team’s numerical simulations.
This research is published in Physical Review Letters.
–Michael Schirber