Not all iron superconductors are the same
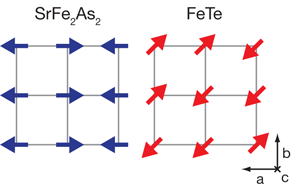
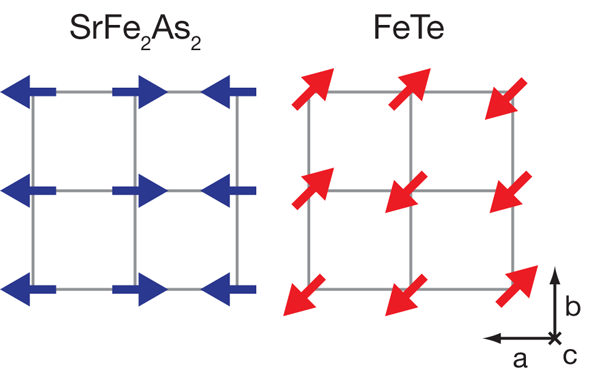
We are facing a new paradigm in superconductivity research with the discovery of superconductivity in iron pnictides ( LaFeAsO, SrFe2As2, BaFe2As2, etc.) and the iron chalcogenides ( FeTe and FeSe). Both classes of iron-based materials, for some as yet unknown reason, have proven to be fertile ground for novel superconductivity, with the transition temperature of the pnictides racing above 50K. A paper published in Physical Review Letters [1] from Yu Qi Xia and colleagues at Princeton University and the Lawrence Berkeley Laboratory, both in the US, in collaboration with Jiao Tong University in Shanghai, and the Institute of Physics of the Chinese Academy of Sciences in Beijing, leads, in concert with other recent experimental work [2,3], to a remarkable conclusion: iron chalcogenides, in seeming contrast to the iron pnictides, do not exhibit the characteristic Fermi-surface-induced magnetism, a spin-density-wave ordering, that the parent compounds of nearly all other iron-based superconductors share (see Fig. 1). The implication for the iron chalcogenides is that the magnetism arises from a different interaction than in the iron pnictides. This also means that in the chalcogenides either the electron pairing that leads to superconductivity and the magnetism arise from different interactions, or that the pairing arises from an interaction distinct from the Fermi-surface-driven itinerant spin fluctuations that are believed by many to create pairing in the iron pnictides.
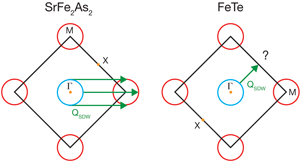
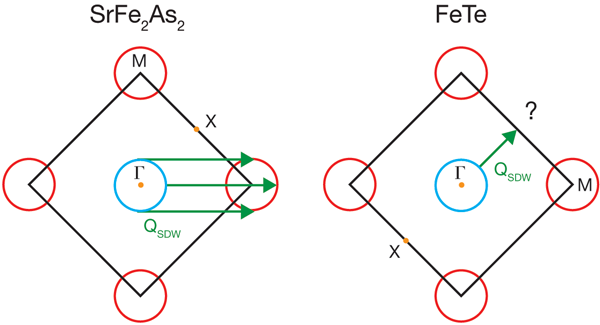
Since the discovery of superconductivity in iron pnictides by Hosono’s group in early 2008 [4], the whole field of superconductivity has been taken by storm. We are now in the exponential growth phase of the field, where the number of preprints appearing on the web numbers over a thousand for the last year. Berndt Matthias’ famous saying “stay away from oxides and magnetic materials” has once again been proven wrong. The discovery of cuprate superconductors clearly indicated the importance of oxide-based materials for achieving higher superconducting transition temperatures. More recently, iron pnictides have proven that magnetic atoms could in fact enable magnetic fluctuations that might play an important role in forming a superconducting state. Now, the next surprise comes from the work of Xia et al. as they provide the first good look into the nature of the ground state of the iron chalcogenide Fe1+xTe. The geometry of the Fermi surface facilitates a phenomenon known as nesting, in which distinct sections of Fermi surface can be made to match via translation by a momentum vector. Their angle-resolved photoemission spectroscopy study reveals that, like virtually all other iron-based superconductors, Fe1+xTe contains the prototypical Fermi-surface structure, with circular hole pockets surrounding the Γ(0,0) Brillouin zone point and elliptical electron pockets centered on the M point, (see Fig. 2). This structure is widely believed to be favorable for spin-density-wave (SDW) nesting, in accordance with the SDW onset experimentally observed between 130 and 190K for essentially all iron arsenide superconductors.
Yet Xia et al. find that the electron and hole Fermi surfaces do not exhibit energy gaps and thus, unlike the parent compounds of nearly all other iron superconductors, show no evidence of a nesting-driven (π,0) instability. Previous experiments [5] find a large electronic contribution to the specific heat, supporting this point of view. Rendering the picture still stranger is the fact that previous inelastic neutron scattering work [2,3] has shown that the ordering vector in Fe1+xTe is (π/2,π/2), which does not connect any Fermi surfaces (see Fig. 2). What is going on here?
One possible explanation is that the magnetism derives neither from localized moments nor Fermi-surface nesting, but simply from the energy gain associated with transfer to lower energy of one-electron density-of-states spectral weight [6]. Another explanation posits a picture of local moments coupled to a low number of itinerant electrons [7] suggesting strong correlation, while a third work [8] postulates an unusual octovalent iron state as creating doping sufficient to generate Fermi-surface nesting at the observed wave vector (although the results of Xia et al.’s study strongly suggest otherwise).
Without commenting on the validity of these explanations, there could be an alternative explanation, as Xia et al. argue: the lack of nesting, combined with the existence of antiferromagnetism, is consistent with a local magnetic moment picture, as suggested in [7]. This is opposed to the itinerant moment scenario.
The suggestion would then be that Fe1+xTe, like the cuprates and unlike other iron-based superconductors, is a more strongly correlated material. Buttressing this point is Xia et al.’s observation of a Curie-Weiss (i.e., 1/T) like behavior in the magnetic susceptibility, suggestive of local moments. However, unlike the cuprates, Fe1+xTe exhibits metallic character even in the presence of magnetism. This material thus forms a “bridge” between the pnictides and cuprates, but with its own distinct metallic character. The experiments are surprising in light of earlier theoretical suggestions of the possibility [9] of an SDW instability in these materials, although more recent theoretical work [6] now matches the experimental magnetic structure.
This material’s behavior has substantial implications for the superconducting state. Does the chalcogenide superconducting state arise from the Fermi-surface structure in which the (π,0) Fermi-surface “ordering” vector leads to a strong spin-fluctuation pairing, as is likely the case in the other iron-based superconductors, or from strong correlations, as is likely the case in the cuprates? Another outstanding question is the role of nanoscale inhomogeneity in iron-based superconductors. Following upon the analogy with cuprates, the presence of local correlations also means that the compound Xia et al. studied is spatially inhomogeneous. Inhomogeneity could be another route to superconductivity, which would form as a result of competing interactions.
The answers to these questions are of profound scientific and practical importance. From a scientific perspective, if the pairing is driven by the electronic instability at the Fermi surface, we may have a case unique to the iron-based superconductors, one in which the pairing and magnetism are driven by different interactions. If the pairing is driven by strong correlations and/or possible inhomogeneity, we may have a different unique case: an iron-based superconductor in which the pairing is not driven by (π,0) itinerant spin fluctuations. Regardless of which case Nature has chosen, it is likely that other chalcogenides can be found in this category. Such an occurrence could reveal a new class of iron-based superconductors. The experience with the LaFeAsO-based family of compounds, in which the maximum Tc quickly doubled from 26K to over 50K in two months, suggests that other chalcogenides with higher Tc may still be found.
From a practical perspective, it is no small point that toxic arsenic is not needed to make the chalcogenides. The only other iron-based superconductors without arsenic content are the phosphides LaFePO and LaNiPO, which both exhibit critical temperatures of 6K or less at ambient pressure. Considering the widespread use of GaAs in the semiconductor industry, the dangers of using arsenic in industrial or scientific applications may be slightly overblown. However, the risk is sufficient that experimental work on the iron-arsenides adheres to very strict guidelines.
The comparison between the chalcogenides and the arsenides leads to the possibility that arsenic, and not simply iron, may be involved in the itinerant properties of most iron pnictides. The discovery of new materials with higher Tc may hinge on this observation and on answering two of the most fundamental questions regarding superconductivity in the iron pnictides: why iron, and why arsenic?
References
- Y. Xia, D. Qian, L. Wray, D. Hsieh, G. F. Chen, J. L. Luo, N. L. Wang, and M. Z. Hasan, Phys. Rev. Lett. 103, 037002 (2009)
- S. Li, C. de la Cruz, Q. Huang, Y. Chen, J. W. Lynn, J. Hu, Y.-L. Huang, F-C. Hsu, K-W. Yeh, M-K. Wu, and P. Dai, Phys. Rev. B 79, 054503 (2009)
- W. Bao, Y. Qiu, Q. Huang, M. A. Green, P. Zajdel, M. R. Fitzsimmons, M. Zhernenkov, S. Chang, M. Fang, B. Qian, E. K. Vehstedt, J. Yang, H. M. Pham, L. Spinu, and Z. Q. Mao, Phys. Rev. Lett. 102, 247001 (2009)
- Y. Kamihara, T. Watanabe, M. Hirano, and H. Hosono, J. Am. Chem Soc. 130, 3296 (2008)
- G. F. Chen, Z. G. Chen, J. Dong, W. Z. Hu, G. Li, X. D. Zhang, P. Zheng, J. L. Luo, and N. L. Wang, Phys. Rev. B 79, 140509 (2009)
- M. D. Johannes and I. I. Mazin, Phys. Rev. B 79, 220510 (2009)
- A. M. Turner, F. Wang, and A. Vishwanath, arXiv:0905.3782
- M. J. Han and S.Y. Savrasov, Phys. Rev. Lett. (to be published); arXiv:0903.2896
- A. Subedi, L. Zhang, D. J. Singh, and M. H. Du, Phys. Rev. B 78, 134514 (2008)