Two Quantum States for the Price of One
Under extremes of pressure and temperature, hydrogen will likely become a superconductor, conducting electricity without resistance. But it may simultaneously become a liquid that flows without friction–a superfluid. This unprecedented combination state, and other related ones, are predicted by computer simulations reported in the June Physical Review B and the 23 September PRL. Whether these unusual states actually occur remains contentious, but experimental tests may soon settle the debate.
Under sufficient pressure–millions of atmospheres, such as occurs in Jupiter’s interior–hydrogen will turn into a liquid metal. At temperatures within a few degrees of absolute zero, it may become a superconductor, in which a current of electrons can flow without resistance. At these temperatures a superconducting state of protons may also emerge [1]. Remarkably, the electron and proton states might cooperate to form a superfluid, in which electrons and protons would move in concert, allowing friction-free motion of mass with no net motion of charge.
Asle Sudbø of the Norwegian University of Science and Technology in Trondheim, in collaboration with two Cornell University physicists, recently predicted some properties of the superconducting superfluid state and two other states–one superconducting but not superfluid, the other superfluid but not superconducting [2]. In their latest two papers, Sudbø and his colleagues describe simulations that explore in detail the interactions of electrons and protons in liquid hydrogen. All three states appear in the simulations, with transitions between them controlled by temperature and an externally applied magnetic field.
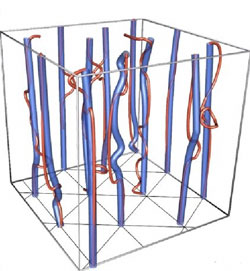
A magnetic field can force many superconductors into a sort of Swiss cheese state, where the field only penetrates through tubes of non-superconducting material called vortex lines, around which electrons circulate. At low temperatures, in the superconducting superfluid phase of liquid hydrogen, vortex lines of both electron and proton superconductors physically coincide and form a fixed lattice.
As you heat the hydrogen, two things can happen, depending on the strength of the magnetic field, the researchers say. In a modest field, the proton vortex lines begin to detach from the electron vortex lines as the hydrogen warms. This separation disrupts the coherent motion of the two components and destroys superfluidity, while superconductivity persists. In their simulations, the team observed the transition to this “electronic superconductor” state.
If the field is fixed at a higher strength as the temperature climbs, the two types of vortex lines cling together, but the composite vortices break out of their rigid lattice positions and move freely, creating what researchers call a vortex liquid. This phase is not a superconductor because an electric current would force the magnetic flux tubes to move, and thus would expend energy. But because the electron and proton charges remain coordinated everywhere, superfluidity is preserved. Sudbø and his colleagues also saw this phase in their simulations.
“The [superconducting superfluid] state would be interesting,” says David Ceperly of the University of Illinois at Urbana-Champaign, but he is skeptical that it can actually occur. The crucial question is whether hydrogen freezes into a solid or remains liquid at low temperatures, and his own computer calculations suggest that it will freeze first. Others, however, disagree [3].
Fortunately, Ceperly and Sudbø agree, experiments may resolve the question before too long [4]. New laboratory methods to make flawless diamonds should soon allow the construction of compression devices that do not fracture at the pressures needed to create metallic superfluid hydrogen. “Only experiments will tell whether or not this state exists,” Sudbø says.
–David Lindley
David Lindley is a freelance science writer in Alexandria, Virginia.
References
- K. Moulopoulos and N.W. Ashcroft, Phys. Rev. Lett. 66, 2915 (1991)
- E. Babaev, A. Sudbø, and N.W. Ashcroft, Nature (London) 431, 666 (2004)
- S. A. Bonev, E. Schwegler, T. Ogitsu, and G. Galli, Nature (London) 431, 669 (2004)
- E. Babaev, A. Sudbø, and N.W. Ashcroft, Phys. Rev. Lett. 95, 105301 (2005)


