A Window into Glass Formation
Window glass and other solids like acrylic and polycarbonate are not crystalline but disordered at the atomic scale–a lot like liquids frozen in time. Researchers have wondered whether they are just very viscous liquids, or whether they are liquids that go through some kind of phase transition as they cool and solidify. Using tiny beads suspended in a liquid as a stand-in for the atoms or molecules in a real glass, a team reports in the 13 July Physical Review Letters that sandwiching the beads between two plates slows them down dramatically, mimicking the cooling of a liquid into a glass. The results strengthen the idea that at least some glasses form when their constituents lock together over long distances in a kind of phase transition.
Window glass and hard plastics are the most familiar examples of a large class of solids called glasses. Many of these materials are very useful, but their atomic-scale behavior is different from traditional crystalline materials and harder to understand. For example, when a glassblower cools molten glass into a solid, its viscosity rises dramatically. For decades, scientists have speculated that this rapid rise in viscosity–known as the glass transition–might hint that the atoms or molecules of a liquid are trying to take on a new arrangement through something like a phase transition. According to this scenario, as the liquid cools, the atoms or molecules must move together in larger and larger clusters that can’t easily slip past each other to let the liquid flow. Researchers have only indirect evidence for these clusters in an ordinary glass, however, in part because available experiments can’t directly observe how the glass’s atomic arrangement differs from that of a liquid.
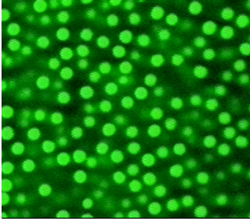
To gain more direct insight into this arrangement, some researchers have used micron-sized plastic particles suspended in a fluid to model atoms or molecules in a glass-forming liquid. Since the beads are much bigger than individual atoms, they are visible under a microscope. A dilute suspension flows like a liquid, but increasing the number of beads makes it act like a paste. When the particles make up more than 60% of the volume, the paste effectively turns into a rigid glass.
To further explore the transition, Eric Weeks of Emory University in Atlanta and his colleagues studied a particle suspension that was enclosed between slightly angled rigid plates that formed a thin, wedge-shaped container. The spacing between the plates varied from nearly zero to about 100 microns. When the group tracked the 3-D motion of the particles over several hours, a clear pattern emerged: the particles moved more slowly where the plates were closer together, even though many particles easily fit between them. “In other words, confinement induces glassy behavior,” says Weeks. When the researchers increased the particle concentration, particle motion was suppressed at a larger spacing, although the team could not go far enough to see the complete rigidity of a glass.
The results show that as particle density increases, the mixture generates larger and larger clusters of particles, according to Weeks and his colleagues. The formation of progressively larger arrangements is the hallmark of the approach to a phase transition.
“The dramatic effect that confinement has on mobility means that particles are no longer behaving as individual entities,” agrees Todd Squires of the University of California in Santa Barbara. Instead, he says, they might be forming small groups of clusters that span the distance between the plates. “This might explain why viscosity suddenly shoots through the roof during the glass transition.”
–Chandra Shekhar
Chandra Shekhar is a freelance science writer in Princeton, New Jersey.
More Information
Physics Today article (requires subscription): J. Langer, “The Mysterious Glass Transition,” Physics Today 60, 8 (2007)