Quartz’s Superficial Attraction
Positronium is a strange sort of atom in which an electron and a positron–the electron’s antimatter partner–orbit one another without a nucleus. Two years ago a team thought they might have created the first positronium molecules, and now they have support from calculations in the 31 August Physical Review Letters. A team of theorists shows how positronium can stick to a solid surface, which is where the experimentalists thought their positronium atoms might be pairing up. The results could also help guide researchers who aim to assemble larger collections of positronium into an exotic quantum state called a condensate.
When physicists shoot a beam of positrons from a radioactive source into a solid target, most of the positrons hit electrons, annihilate, and form gamma rays. However, some of the positrons are able to survive and pair up with electrons as positronium, either at a surface or inside the solid, and then fly out of the material.
Measuring the lifetime of this atom–roughly 100 nanoseconds–in a porous material can provide detailed information about the pore sizes because the pore surfaces strongly influence positronium’s creation and decay. In a related measurement in 2005, a team led by Allen Mills of the University of California at Riverside reported short lifetimes for high-density positronium in nano-porous silica–a non-crystalline form of quartz (see 2005 Focus story). One explanation, they suggested, was that pairs of positronium atoms were forming short-lived molecules at the pore surfaces–possibly the first observation of such matter-antimatter molecules.
But theorists don’t fully understand positronium’s interaction with a surface. For example, despite past experimental evidence that positronium sticks to the surface of a quartz crystal, the only detailed theoretical work to date, from 30 years ago, found that sticking was impossible [1].
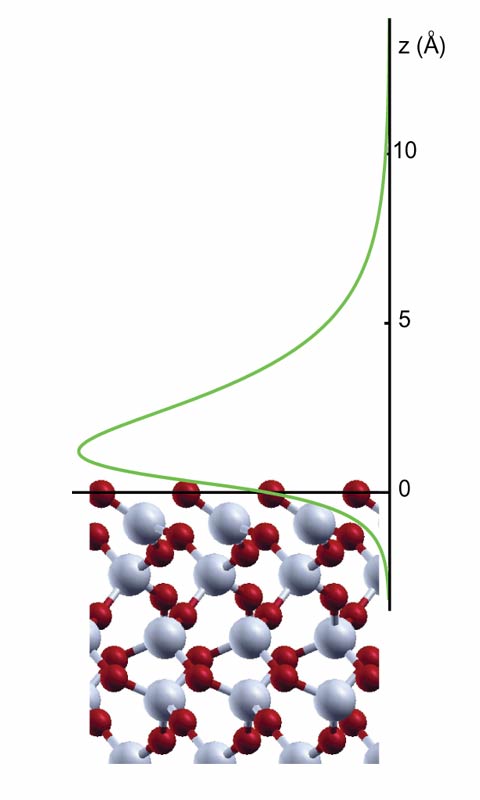
Since that time, however, “the theoretical tools have evolved a lot,” says Rolando Saniz of Northwestern University in Evanston, Illinois. He and his colleagues have now used modern techniques to better calculate the effects on quartz caused by oscillating electric fields of a range of frequencies. Oscillating fields coming from random fluctuations in the positronium atom and from quartz atoms are an important part of the so-called van der Waals attraction between positronium and the quartz surface. The positronium is repelled if it gets too close, however, because there are no empty quantum states in the quartz for its electron. Combining these elements, the team calculates that positronium binds near the surface with an energy of 0.14 electron-volts, in close agreement with the experiments that showed positronium sticking.
The team says that once two positroniums are trapped on a surface, they can easily combine to form a dipositronium molecule. The surface not only keeps the particles near each other after they collide; it also absorbs and imparts momentum through a multi-step process that allows the pairing to occur.
Mills says the results support his team’s suggestion that it created dipositronium two years ago. “Now we have more confidence in this possibility, because of Saniz et al.’s calculation indicating the existence of a positronium surface state.” He thinks that at higher densities the trapped positronium could collect into a Bose-Einstein condensate (BEC), a quantum macro-molecule previously observed in ultra-cold gases. Physicists are keenly interested in a positronium BEC, in part because it could power a gamma-ray laser. However, Mills cautions that positronium trapped at surfaces may disrupt laser operation, so experimenters may need to find a way to disable this trap by perhaps altering the surface makeup.
–Michael Schirber
Michael Schirber is a Corresponding Editor for Physics Magazine based in Lyon, France.
References
- G. W. Ford, L. M. Sander, and T. A. Witten, “Lifetime Effects of Positronium in Powders,” Phys. Rev. Lett. 36, 1269 (1976)
More Information
A 2005 Focus story describes the apparent formation of dipositronium.