Bacteria Give Stirring Performance
Like flocking birds and swarming locusts, swimming bacteria will coordinate their motion when highly concentrated. A team has now developed a technique that can capture a three-dimensional snapshot of the density of these cooperating bacteria inside a thin film of fluid. The results, reported in the September Physical Review E, show the rising and falling of bacterial plumes, which may help to increase the bacteria’s access to oxygen. The work could improve understanding of cooperative bacterial behavior and could also help researchers design better micro-mixers.
Collective motion has been observed throughout biology from sperm cells to wildebeests. Swimming bacteria, such as Escherichia coli and Bacillus subtilis, have been especially challenging to explain. Interactions among the microbes and the fluid enable the bacteria to form swirling vortices and dense congregations that span ten times the size of the individuals. But the exact mechanisms that drive this synchronized swimming are not fully understood, in part because the motion has only been seen in two dimensions from imaging of the fluid surface.
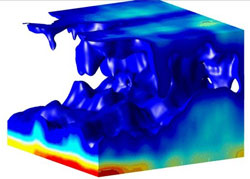
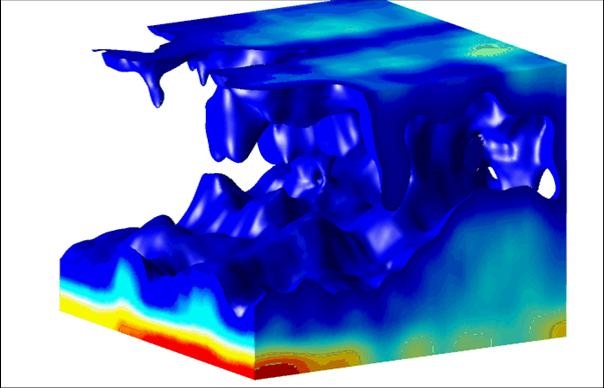
Igor Aranson of Argonne National Laboratory in Illinois and his colleagues have now designed an imaging technique that can observe dense bacterial populations in three dimensions. In this first demonstration they viewed B. subtilis swimming in a thin film of fluid stretched between 4 crossed wires that formed a square frame. The density of bacteria in the film was around per cubic centimeter, approximately 20 times denser than their normal state. By changing the size of the square, the team could indirectly vary the thickness of the film. With a conventional microscope they observed dense bacterial clusters visible at the surface when the film thickness was over 200 microns. These groupings were about a hundred microns wide and lasted for about 7 seconds.
To explain this collective motion, the team used optical coherence tomography (OCT), a technique that has been used in areas such as medical imaging and art restoration and is similar to ultrasound imaging. A light beam hits a point on the sample, and the backscattered (diffusely reflected) light is compared with a reference reflection from a movable mirror. The interference of the two determines the density of scatterers (in this case bacteria) at a specific depth. The system builds up a three-dimensional image by using many mirror positions for each sample point and scanning across many points on the sample surface. The researchers fit the density maps with a model that incorporates the diffusion of bacteria and oxygen.
The results showed that most of the B. subtilis swim along the top and bottom surfaces, where they occasionally bunch together. They gobble up oxygen diffusing into the fluid, and if the film is thick enough, the middle layer becomes highly depleted in oxygen–so much so that any bacteria that happen to dive away from the top surface will not have the energy to swim back to the surface. Instead, they will fall due to gravity and pull some of their neighbors along in the flow. Some bacteria on the bottom surface get swept upward by the circulating flow that develops. The resulting plumes of “out of breath” bacteria show up as persistent dark spots on the surface.
Rising and sinking bacterial flows have been seen before in B. subtilis, but only in much thicker films and more dilute solutions, where each cell has little influence on its neighbors. The research team found that the coordinated movement in their thin films helps to mix both the bacteria and oxygen ten times faster than would occur if individual bacteria were moving randomly. The authors speculate that this enhanced mixing could help the bacteria survive harsh conditions, such as a shrinking water environment due to evaporation. “If they did not do something like this, they would likely die,” Aranson says.
The new experiment shows that convective mixing occurs in a wider range of circumstances than previously thought, says Eric Lauga of University of California, San Diego. So the authors think their results could benefit research on improving mixing in micro-sized fluids using either real bacteria or machines that mimic them.
–Michael Schirber
Michael Schirber is a Corresponding Editor for Physics Magazine based in Lyon, France.
More Information
Synchronized Swimming in Bacteria (Physics News Update, 2004)
Movies of Swimming Bacteria ((Rowland Institute at Harvard)