Imaging the Heart’s Blood Supply
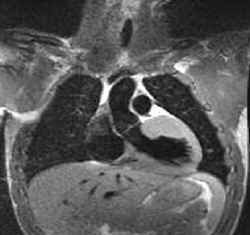
Most heart attacks are caused by a narrowing of the heart’s blood vessels, which reduces the oxygen supply to the muscle. A German team is now developing a method that uses magnetic resonance imaging (MRI) to diagnose the effectiveness of those arteries. Their technique–an improvement on previous attempts by others–is based on the small differences in magnetic field generated by deoxygenated blood compared with the oxygen-carrying form. As part of that work, the team describes a new theory in the 15 November PRL relating magnetic resonance data with the properties of blood flow in the heart.
In this MRI method, the spins of water protons throughout a patient’s body are lined up in a large magnetic field, and a pulse of radio waves is applied which tilts the spins away from their alignment. The machine then detects the protons’ radio wave responses as each proton’s spin precesses (rotates) rapidly at a rate determined in part by the precise value of its local magnetic field. The protons’ signals quickly begin to cancel one another because they experience slightly different local fields which cause them to precess at different rates. The time until cancellation, called is shortened by the presence of deoxyhemoglobin (hemoglobin without oxygen bound) in nearby capillaries because the iron atoms create a widely varying magnetic field. The oxygen-carrying form of hemoglobin is not magnetic, so oxygen-starved regions of the heart should have shorter times.
In the early 1990s researchers began exploiting this principle to track blood flow in the brain with so-called functional MRI, but the heart muscle’s motion and varying blood supply have made it a more difficult target for the technique. The German team, led by Wolfgang Bauer of the University of Heidelberg and Lothar Schad of the German Cancer Research Center in Heidelberg, has developed the technique to the point that they can generate detailed maps of throughout the heart and see clear differences between healthy patients’ hearts and those of patients with restricted blood flow. But to properly interpret the data, the team needed a good theory. “In the heart there has been no real analytical model,” says Bauer; instead, others have relied on computer simulations, he says.
One difficulty of the data analysis is that protons experience different magnetic fields based on two separate effects, both of which contribute to in the heart: First, at the moment of the radio pulse, the location of each proton in the heart tissue has a different magnetic field; and secondly, as the protons diffuse through cells, they sample many locations during the time The new theory tries to account for both effects and relates capillary blood volume and oxygen level to The theory gives values in agreement with the computer simulations of others and with the team’s own data on real patients.
“Their approach is a novel one,” says John Forder of the University of Alabama at Birmingham. But he is concerned that the German team has not yet solved the biggest problem faced by others: the difficulty of distinguishing between a change in blood volume supplying a region of the heart and a change in deoxyhemoglobin concentration–both of which affect Bauer says the new theory will help his group address that problem properly. As for the future of the technique, “I have high hopes for it,” says Forder.


