Out of the substrate, an atomic chain
The ability to accurately and reproducibly create nanostructures with a known composition and set of properties is a fundamental goal in nanoscience. In addition to the lithographic techniques usually employed in this pursuit, self-assembly or self-organization provides an attractive route towards creating nanoscale objects.
The self-assembly of atoms and molecules deposited on a surface is guided by the competing mechanisms of diffusion, aggregation, and intermixing. However, if intermixing is dominant during the deposition process, the nano-objects that form can be composed of the substrate material instead of the deposited material.
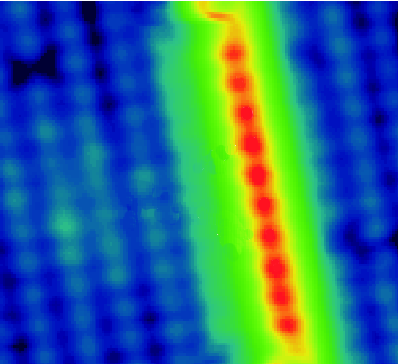
Using scanning tunneling microscopy and spectroscopy, D. H. Wei, Chunlei Gao, Kh. Zakeri, and Marek Przybylski at the Max-Planck-Institut für Mikrostrukturphysik in Halle, Germany, report in Physical Review Letters that whether they deposit a submonolayer of chromium, manganese, iron, cobalt, or nickel on a palladium surface, the metal chains that ultimately form are made of palladium. Gao et al. argue that for the particular palladium surface they have chosen to study, intermixing is more energetically feasible than surface diffusion because of the large lattice mismatch between the substrate and the deposited material and they theorize that the deposited atoms simply diffuse into the bulk.
This solves the puzzle of why the scanning tunneling spectra of the atomic chains on this surface showed the same features regardless of the deposition material, and emphasize the role of atomic intermixing in self-assembly of nanostructures. – Daniel Ucko