Inducing Multiple Reactions with a Single Photon
Many fundamental processes in nature, such as photosynthesis or human vision, are due to chemical reactions that are triggered by light. In a simplified picture of such processes we assume that photons merely provide the necessary energy to the chemical system to overcome a certain reaction barrier. Besides this energy contribution, the photons supposedly do not influence the system any further. Such a perturbative picture of the interaction between light and matter is reasonable in many situations of photochemistry and justifies what is known as the Stark-Einstein law [1]: for each absorbed quantum of light, no more than one molecule undergoes a chemical reaction.
Nature, however, does not need to adhere to this law and can violate it in specific cases. Javier Galego from the Autonomous University of Madrid and co-workers [2] consider such a case, in which a handful of molecules are strongly coupled to a confined light mode in an optical cavity [3]. The authors predict that a single photon entering the cavity can set off a reaction involving many more than just one molecule. What makes this example so special is that on the one hand it is simple—not needing specialized resonance conditions as do normal chain reactions—and on the other hand it highlights the complexity of chemical reactions, which are defined by an intricate interplay among electrons, nuclei, and photons. If such multimolecule triggering can be realized, it could have important practical implications in solar energy storage. Molecules that under normal conditions efficiently absorb solar energy could be brought into strong coupling so that a single photon could release the stored energy of the whole ensemble on demand.
The key difference between Galego et al.’s work and previous studies of photochemistry is that the authors consider a situation where the standard assumption that the photons are merely an external perturbation is no longer justified. The electromagnetic field associated with the cavity modes becomes part of the chemical system and can change its properties. This happens by an effect called hybridization, where the matter degrees of freedom become mixed with, or dressed by, photons. The combined states of light and matter states are called polaritons [3, 4]. Galego and collaborators investigate polaritonic chemistry in the special case where the hybrid light-matter states arise by merely coupling to the vacuum of an “empty” cavity.
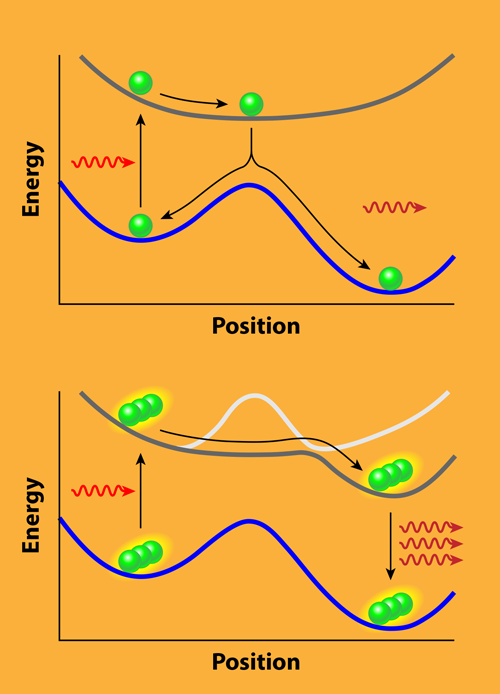
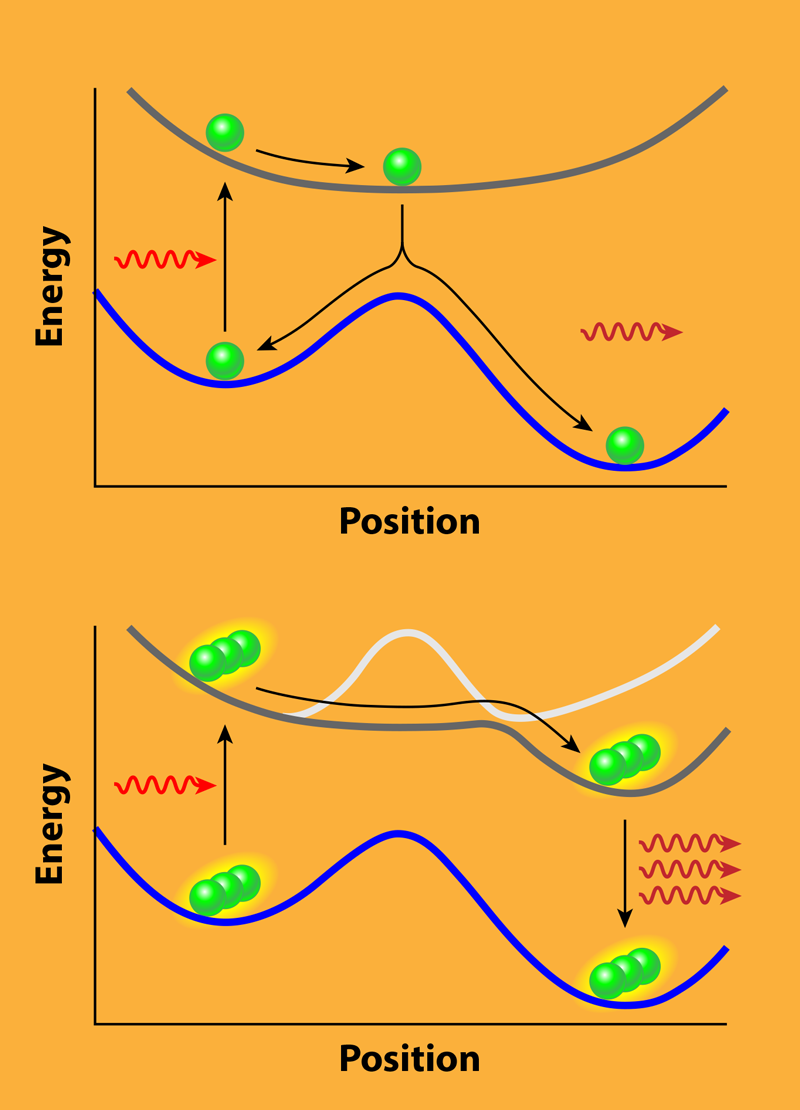
The authors start by considering a “bare” molecule, which is a molecule outside of the cavity. They use a simple model in which the molecule has a metastable state with a large activation barrier separating it from the minimum-energy ground state. This barrier leads to a very long lifetime for the metastable state, making the system interesting for energy storage. One way to release this stored energy is to excite the molecule from the metastable state to an excited state. If the molecule relaxes to the ground state, the stored energy can be recovered as a photon. The challenge is that launching this reaction may require many photons, and even with sufficient excitation, the molecule may just relax back to the metastable state, giving this energy release process an overall low yield.
Upon placing an ensemble of these molecules inside a cavity, the coupling to the cavity mode changes the bare potential-energy surfaces. These surfaces, which define how the molecules move relative to each other, are a good indicator for how chemical reactions will proceed. While the ground-state surface that contains the reaction barrier stays basically unaltered, the excited-state surfaces change significantly when the molecules enter the cavity (see Fig. 1). For the case in which the cavity mode is in resonance with the electronic excitation at the metastable state, the quantum yield of releasing the energy upon absorbing a single photon is enhanced. More importantly, the cavity mode couples to all molecules of the ensemble at the same time. It therefore entangles the molecules and leads to the formation of a “supermolecule.” This is the reason that the absorption of a single photon by one of the molecules can induce a chain reaction, where one molecule after another releases the energy stored in its metastable state.
The results predict experimentally accessible effects that one could look for in a setup, as in Ref. [5], where the target molecules are placed in a host material and then sandwiched between two mirrors. Of course, the simplified theoretical modeling disregards many details that can become important in an actual experiment. The authors, for example, do not explicitly consider decoherence and dissipation effects, nor the interference between the nuclear wave packets, which characterize the nuclear motion. Moreover, strong coupling can have significant impact on the actual details of real molecules. Here, ab initio methods that fully treat the couplings between the molecule and the cavity field [6] can be used to scrutinize the findings by Galego and co-workers and provide a more realistic description of the experimental observables.
Besides the potential applications of single-photon triggering in, for example, stored energy retrieval, there is a second, more fundamental aspect that merits mentioning. In normal chemistry, we tend to make a distinction between light and matter, but polaritonic chemistry shows that this is just a convention. In the bare case, what we consider to be “the molecule” is always dressed by photons and therefore defined with respect to the electromagnetic field of the vacuum. Changing the vacuum with an optical cavity redefines “the molecule,” as the surrounding electromagnetic field fluctuations are different. This picture is not so surprising if one remembers that in quantum electrodynamics, the physical electron is always considered as a “bare” object dressed by the photon field. But it is still astonishing that without highly relativistic energies or high-precision measurements [7] quantum-field effects become accessible in chemistry. The collective interaction and dressing of molecular states can provide novel insights into how photons interact with charged particles. Thus by developing applications based on the hybrid nature of polaritonic states, we also learn more about the fundamental description of light-matter interaction.
This research is published in Physical Review Letters.
References
- K. K Rohatgi-Mukherjee, Fundamentals of Photochemistry, 3rd ed. (New Age International, New Delhi, 2014)[Amazon][WorldCat].
- J. Galego, F. J. Garcia-Vidal, and J. Feist, “Many-Molecule Reaction Triggered by a Single Photon in Polaritonic Chemistry,” Phys. Rev. Lett. 119, 136001 (2017).
- T. W. Ebbesen, “Hybrid Light-Matter States in a Molecular and Material Science Perspective,” Acc. Chem. Res. 49, 2403 (2016).
- J. Flick, M. Ruggenthaler, H. Appel, and A. Rubio, “Atoms and Molecules in Cavities, from Weak to Strong Coupling in Quantum-Electrodynamics (QED) Chemistry,” Proc. Natl. Acad. Sci. U.S.A. 114, 3026 (2017).
- J. Hutchison, T. Schwartz, C. Genet, E. Devaux, and T. W. Ebbesen, “Modifying Chemical Landscapes by Coupling to Vacuum Fields,” Angew. Chem. Int. Ed. Engl. 51, 1592 (2012).
- M. Ruggenthaler, J. Flick, C. Pellegrini, H. Appel, I. V. Tokatly, and A. Rubio, “Quantum-Electrodynamical Density-Functional Theory: Bridging Quantum Optics and Electronic-Structure Theory,” Phys. Rev. A 90, 012508 (2014).
- S. G. Karshenboim, Precision Physics of Simple Atoms and Molecules, Lecture Notes in Physics Vol. 745 (Springer, Berlin, 2008)[Amazon][WorldCat].