A Coordinated Approach to Chemical Reactivity
Atoms are gregarious by nature, escaping a solitary existence by forming chemical bonds with their neighbors. Chemical reactivity, in turn, depends on the degree of atomic “isolation”; a lonely atom will more readily interact and form bonds with others, as compared to its more “coordinated” counterparts that are chemically engaged with other atoms. This coordination concept plays a central role in our understanding of chemical and catalytic activity. What has been lacking to date, however, is a direct and quantitative measurement of the variation in chemical reactivity of atoms having different degrees of coordination. This is precisely what Julian Berwanger at the University of Regensburg, Germany, and colleagues have now achieved [1]. The Regensburg team probed how the neighborhood of an atom affects its propensity to react with a carbon monoxide (CO) molecule, which they guided into position around an iron cluster with subangstrom precision. Their results quantify how the reactivity of atoms of iron (Fe)—an important material for catalysis—varies as a function of the atoms’ coordination.
Surface scientists have long recognized the importance of the coordination number c—a measure of the number of chemical bonds formed by an atom. Crack a crystal apart to expose a surface, and those atoms that were previously in the bulk are wrenched from their neighbors, lowering their coordination numbers and producing what are known, rather descriptively, as dangling bonds. The complexity deriving from this reduction in symmetry is part of the reason why Pauli famously described surfaces as being “invented by the devil.” Some parts of a surface, however, can be almost perfectly flat, with atoms that have lost their neighbors above but are still largely among “friends.” These atoms have a relatively high coordination number, and correspondingly low chemical reactivity. Other atoms, by contrast, find themselves in an environment of even lower symmetry—at atomic steps on an otherwise flat surface or at the edges of a nanoscopic cluster, for example—which enhances their chemical reactivity [2]. Studies of catalytic materials have established, for instance, that step edges are the most active catalysis sites and that catalytically inactive surfaces can be activated by adding single atoms or atom clusters to the surface.
Recognizing that a systematic approach to quantifying the role of coordination requires exceptionally well-defined atomic clusters, Berwanger and colleagues exploited a scanning probe microscope (SPM) to achieve the ultimate control in nanostructure fabrication: they built, from the bottom up, two-dimensional Fe clusters, comprising from 3 to 15 atoms, and used the same SPM to image the atoms and probe their reactivity.
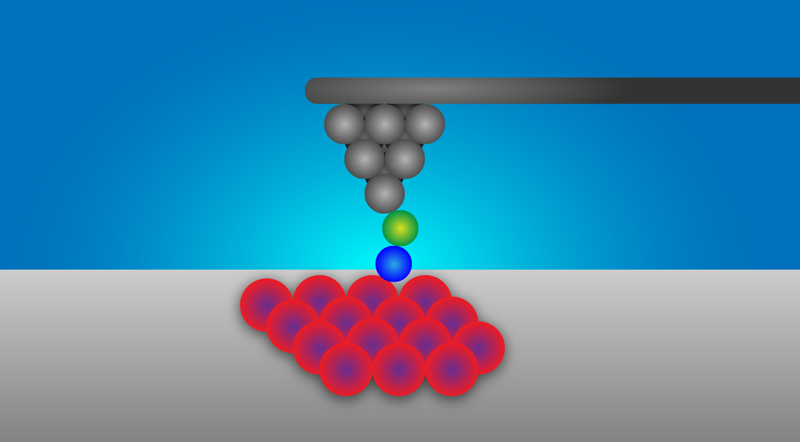
SPM images a sample by probing it with an atomically (or molecularly) sharp tip. Several tip-sample interactions are possible, including the quantum-mechanical tunneling of electrons exploited by a scanning tunneling microscope (STM). The setup used by the Regensburg team relies on detectable shifts in the oscillation of a cantilever (which holds the tip) due to chemical interactions between the apex of the tip and the sample atoms (see Fig. 1).
The spatial resolution of this variant of SPM—known as noncontact atomic force microscopy (NC-AFM) or dynamic force microscopy—is such that it can image, manipulate, and spectroscopically analyze not just single atoms but single chemical bonds. The researchers’ setup, in particular, employs two ingredients that boost SPM resolution. The first is a tuning-fork sensor operated in the so-called qPlus mode, which was pioneered by the team’s leader Franz Giessibl and is at the heart of the majority of today’s ultrahigh resolution NC-AFM setups [3]. The second is the functionalization of the tip through the attachment of a CO molecule—a protocol introduced a decade ago. [4].
In SPM, the very same probe that is used to acquire an image can also be applied in a more invasive fashion: it can push, pull, prod, and poke single atoms and molecules. This type of atomic manipulation was pioneered by Don Eigler and Erhard Schweizer at IBM Almaden thirty years ago with their atom-by-atom construction of the IBM logo [5], which inspired a series of ever-more impressive demonstrations of atomic-level control of condensed matter (as described in a recent review [6]).
Building on these earlier efforts of atomic control, the Regensburg team utilized their tip to build 2D clusters comprising between 3 and 15 iron atoms, systematically controlling the degree of coordination of the atoms. They then used their CO-topped tip to probe specific iron atoms in the differently sized clusters. As they neared the STM tip to individual Fe atoms, a chemical bond formed with the CO molecule at the tip apex. By accounting for variations in the bonding of the molecule to the tip, the team could then extract the strength of the bonding between the CO and Fe atoms of varying coordination number. The bond strength of the Fe-CO bond is a direct indication of the atom’s reactivity: the stronger the bond, the more reactive the atom.
The experiments showed that the coordination number had a profound impact on chemical reactivity. The least coordinated atoms at the corners ( c=2) and at the edges of the cluster ( c=3 to 4, depending on cluster size) were chemically much more reactive than atoms inside the cluster ( c=5 to 6). While the behavior was intuitively expected, the experiments provide the first quantitative determination of chemical reactivity as a function of coordination at the single-atom level.
The approach introduced by Berwanger et al. has the potential to be adopted across a wide range of fields—surface science, nanoscience, and condensed-matter physics and chemistry. Since the CO tip functionalization is routinely used in high-resolution scanning probe microscopy, it should be relatively straightforward to adopt the protocol laid out by Berwanger and colleagues. A key limitation is that their approach is presently limited to 2D clusters. This difficulty is inherent to SPM, but researchers are investigating pathways to move beyond “flatland” and explore 3D structures [7], including the use of machine-learning techniques to mine SPM data [8, 9].
It’s almost verboten now to discuss advances in nanoscience and nanotechnology without invoking Richard Feynman’s prescient 1959 speech “There’s Plenty of Room at the Bottom” [10]. Berwanger et al.’s study instead brings to mind a less celebrated, though no less important, thought from Feynman: “What I cannot create, I do not understand.” By creating atomically precise clusters and analyzing their creations with single-bond precision, the Regensburg team has delivered a deeper understanding of the impact of an atom’s neighborhood on its chemical reactivity.
This research is published in Physical Review Letters.
References
- J. Berwanger et al., “Atomically resolved chemical reactivity of small Fe clusters,” Phys. Rev. Lett. 124, 096001 (2020).
- A. Wang et al., “Heterogeneous single-atom catalysis,” Nat. Rev. Chem. 2, 65 (2018).
- F. J. Giessibl, “The qPlus sensor, a powerful core for the atomic force microscope,” Rev. Sci. Instrum. 90, 011101 (2019).
- L. Gross et al., “The chemical structure of a molecule resolved by atomic force microscopy,” Science 325, 1110 (2009).
- D. M. Eigler and E. K. Schweizer, “Positioning single atoms with a scanning tunnelling microscope,” Nature 344, 524 (1990).
- A. A. Khajetoorians et al., “Creating designer quantum states of matter atom-by-atom,” Nat. Rev. Phys. 1, 703 (2019).
- C. Moreno et al., “Imaging three-dimensional surface objects with submolecular resolution by atomic force microscopy,” Nano Lett. 15, 2257 (2015).
- O. Gordon and P. Moriarty, Mach. Learn.: Sci. Technol. (to be published).
- B. Aldritt et al., “Automated structure discovery in atomic force microscopy,” arXiv:1905.10204.
- http://www.phy.pku.edu.cn/~qhcao/resources/class/QM/Feynmans-Talk.pdf.