Instant-Freeze Water
On Earth, there’s mostly one type of ice—the hexagonal crystal found in glaciers and snowflakes. Laboratory experiments, however, have shown that ice may come in other phases. Ice VII, for instance, is a cubic crystal that can form under high pressure even at hundreds of degrees Celsius. Researchers have speculated that this “hot ice” might also be found on the ocean floor of Saturn’s moon Titan or on other exoplanets containing water. Now, Arianna Gleason of Stanford University, California, and Los Alamos National Laboratory, New Mexico, and co-workers have successfully observed the real-time growth of ice VII that they formed under laser-induced pressure shocks. Their findings may help scientists refine thermodynamic models for solid-liquid phase transitions.
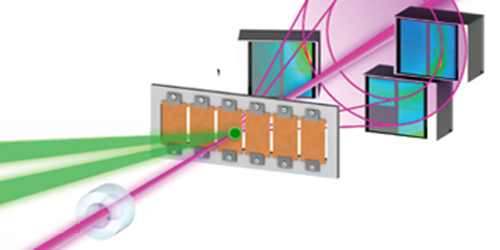
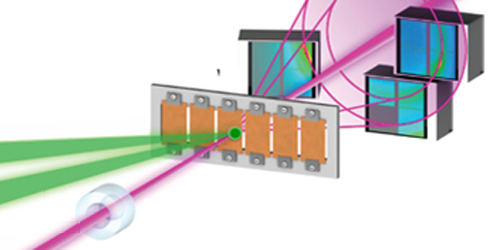
Other groups have, like Gleason and her colleagues, attempted to “shock freeze” water, using lasers to create pressure changes. These studies, however, could not characterize the structure of the phase that is quickly generated after laser illumination. The authors achieved this feat using the ultrashort pulses from the x-ray free electron laser (XFEL) of Stanford’s Linac Coherent Light Source. In each experimental cycle, an infrared laser pulse vaporized the surface of a thin water layer, creating shock waves that compressed the liquid. The XFEL x-ray pulse arrived at a controllable time after the infrared pulse and was diffracted by the sample.
Within 6 ns of the laser shock, the team detected two diffraction peaks that unambiguously revealed the formation of crystalline ice VII. Based on fits of the temporal dynamics of the time-varying diffraction peaks, the team argues that the freezing water first crystallizes into 1D structures like needles and rods, which then nucleate to form solid ice VII.
This research is published in Physical Review Letters.
–Matteo Rini
Matteo Rini is the Deputy Editor of Physics.