Chemistry Drives Convection
Stirring a mixture can accelerate chemical reactions between the ingredients, but less obvious is the reverse: reactions can stir up a mixture. As a demonstration of the chemistry “tail” wagging the fluid dynamics “dog,” researchers have run several computer simulations and verified their model with a simple acid-base experiment. Described in the 29 January Physical Review Letters, the results explicitly show how common chemical reactions can induce fluid flows. Such chemically-driven fluid motion may have important effects in geology, astrophysics, and pollution control.
Convection occurs when lower density fluid is located below higher density fluid–the lower density material rises, and the higher density material sinks. The best known case is where the lower density fluid is warmer, but it need not be. Since the 1980s researchers have been studying convection triggered by “autocatalytic” reactions, which are self-promoting. But there has been little study of the effects of more common chemical reactions on fluid flow, which could be relevant to many areas of science, such as the geology of Earth’s mantle.
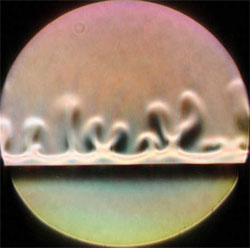
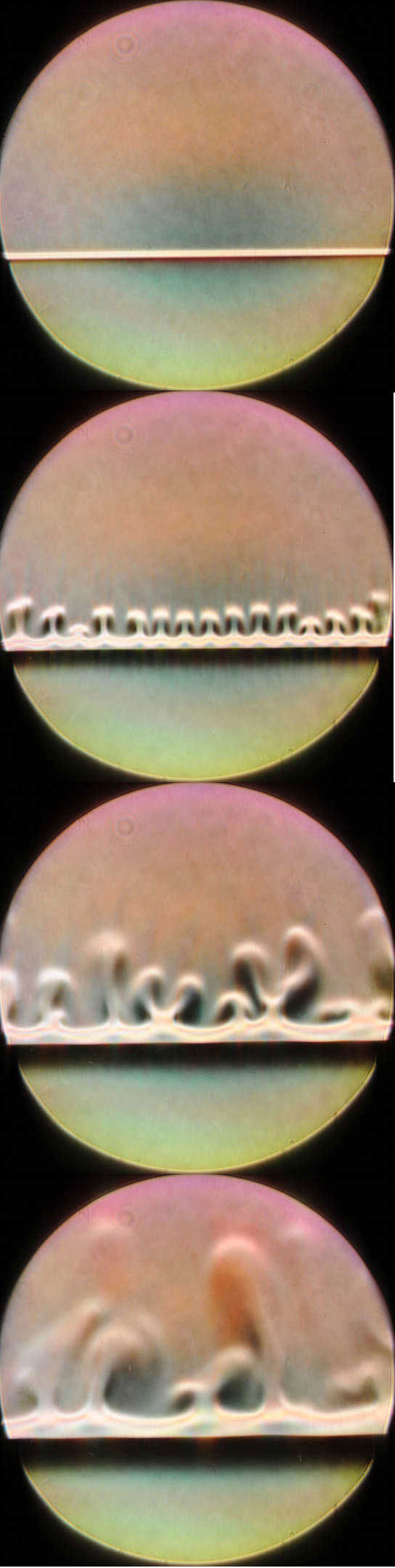
Anne De Wit and her colleagues at the Free University of Brussels (ULB) looked at the general case where two reactants come together to produce a single product (A+B→C). They developed a hydrodynamic model and then performed several computer simulations in which a less dense solution containing reactant A was placed on top of a more dense solution with reactant B. This normally stable configuration was disturbed by the appearance of the product C at the boundary between the two solutions, which led to convective “fingers.”
Although this chemically-induced convection was not a surprise, the convection patterns in the simulations were surprising. In some cases the convective plumes went only upward from the boundary, rather than creating a symmetric pattern that sent fingers both up and down from the mid-line.
It turns out that even without a chemical reaction, convection would be expected if the reactants had different diffusion rates. Here, diffusion is the spreading of dissolved atoms or molecules from regions of higher concentration to regions of lower concentration, without an accompanying flow of the entire fluid. Diffusion differences can lead to density variations that create convection, but without a chemical reaction, the convection would be symmetric.
To verify their simulations, the team observed hydrochloric acid (HCl) and sodium hydroxide (NaOH) reacting to form water and table salt (NaCl, which remains dissolved in the solution). Using a thin chamber between two vertical glass slides, they slowly injected a solution of HCl from the top and a relatively dense solution of NaOH from the bottom. Variations in the fluid density were detected by sending laser light through the experimental cell. This technique is less invasive than the one used in an earlier experiment looking at the same reaction [1] says De Wit’s coauthor Christophe Almarcha *.
As the simulations predicted, the team saw convection plumes only in the top half of the cell. HCl diffuses faster than the product NaCl (sodium and chloride ions). Consequently, downward-diffusing HCl is not compensated fast enough by upward-diffusing NaCl in the region just above the boundary. The partial vacancy that results is less dense than the fluid above it, so convection ensues. Nothing similar occurs below the boundary because NaCl and NaOH have similar diffusion rates.
The new simulations show that convection makes the reactions progress faster, just as stirring would. This could mean that researchers have underestimated reaction and mixing rates in natural phenomena, like the Earth’s mantle and supernova explosions, as well as for carbon sequestration, in which carbon dioxide is stored underground. Modelers have so far not considered how convection induced by the interaction of carbon dioxide with ground water may affect the long-term containment of this greenhouse gas, says De Wit. “Our work shows you can’t neglect these reactions,” she says.
“The authors do a nice job of demonstrating, both numerically and experimentally, in a very simple system, how chemistry (an acid-base reaction) and physics (convection) can be inextricably intertwined and lead to new spatiotemporal phenomena,” says Irving Epstein of Brandeis University in Waltham, Massachusetts. “The present paper carries the theoretical analysis further than earlier work and presents some very pretty experiments.”
* Almarcha says the authors of [1] tracked density changes with a tracer dye, and the tracer had an unexpected influence on the fluid motion, leading to very different results. They also did not come up with a complete theory and simulations of the fluid flow.
–Michael Schirber
Michael Schirber is a Corresponding Editor for Physics Magazine based in Lyon, France.
References
- A. Zalts et al., “Pattern Formation Driven by an Acid-Base Neutralization Reaction in Aqueous Media in a Gravitational Field,” Phys. Rev. E 77, 015304(R) (2008)
More Information
Chemical Plumes–description and video of convection from autocatalytic reactions