Mixing it Up (Or Not)
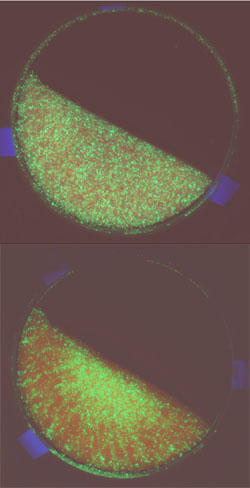
If you fill a barrel part-way with red beads, add some green beads, and then roll it around the room a bit, will your beads blend? It depends on the properties of the two bead types, but conventional wisdom holds that wet beads will remain mixed more easily than dry ones. In the 9 May PRL, a team shows that this thinking is not always correct. By mixing wet and dry beads in a tumbler, and developing a theory for interacting beads, they found the precise conditions that lead to mixing or separation. In some cases, simply adding water will cause two types of particles to separate. Their theory might suggest improved processing methods in many industries, from pharmaceutical manufacturing to fuel delivery in power plants.
Although similar to liquids, flowing granular mixtures–like beads in a barrel–have behaviors all their own. The different components sometimes mix or unmix themselves in ways that are still poorly understood. Physicists still don’t know how to prevent cereal dust from settling to the bottom of the box, for example. But milk makes the dust stay glued to the cereal flakes, so sticky or cohesive mixtures behave differently from dry ones.
Cohesion in a granular mixture could come from a fluid like water, or from the weak attraction among molecules called the van der Waals force. Unfortunately, cohesion is hard to put into a theory. “Most real problems involve some cohesion, while most models neglect it” because of the complications, explains Troy Shinbrot of Rutgers University in Piscataway, New Jersey. Models that do account for cohesion often describe it as an average property, identical for all particles in the mixture.
Hongming Li and Joseph McCarthy of the University of Pittsburgh took the opposite approach. They developed a mathematical expression for the force acting between any pair of particles, accounting for size, density, and each particle’s affinity for water. To predict whether two types of particles would mix or separate, the team compared the strength of attraction between pairs of identical particles with that of mismatched pairs. If the mismatched pairs had higher attraction, they predicted thorough mixing; otherwise, they expected the particles to segregate.
To test the predictions, Li and McCarthy filled their disk-shaped tumbler half-way with colored beads with different properties. They varied the densities, sizes, and surface properties of the particles and tested each combination dry and then wet. As the theory predicted, under some conditions, the dry particles mixed, but adding water caused them to separate. In other cases, water forced otherwise segregating particles to blend.
The explanation of the water’s effects lies in the beads’ surface properties. If the red beads are water loving (hydrophilic) and the green beads have water-repelling (hydrophobic) surfaces, adding water will affect them differently. The red beads will tend to clump into wet clusters, while the green beads will avoid them. On the other hand, if the beads are of two different sizes, but both hydrophilic, the dry mixture will separate–think of the cereal box–but water can act as a glue, with the smaller particles sticking to the larger ones.
“Making a granular material wet adds a new intergrain interaction which is qualitatively different from anything in dry grains,” says Peter Schiffer of Pennsylvania State University in University Park. He says the new work shows the important effect of this interaction on mixing of the whole system. “These results are of great fundamental interest, but they also should be of practical importance” for industrial processing, he says.
–Kim Krieger
Kim Krieger is a freelance science writer in Norwalk, Connecticut.


