X-Rayed Movie
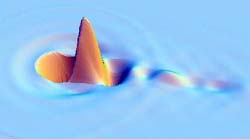
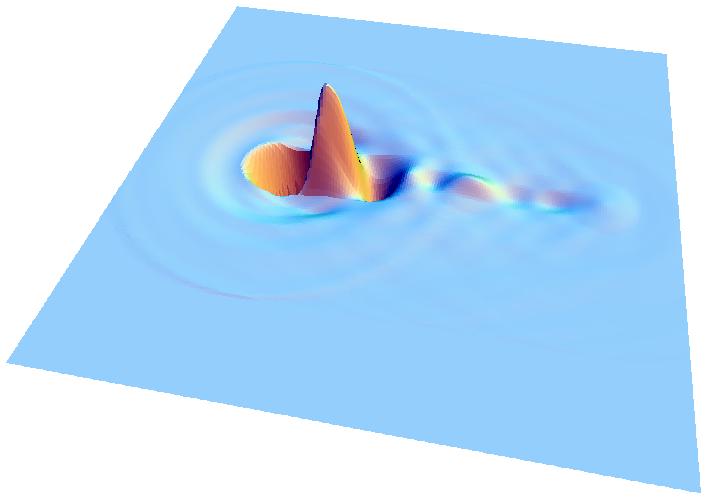
A research team has produced the fastest movies ever made of electron motion. Created by scattering x rays off of water, the movies show electrons sloshing in water molecules, and each frame lasts just 4 attoseconds (quintillionths of a second). The results, published in the 11 June PRL, could let researchers “watch” chemical reactions even faster than those viewable with today’s “ultrafast” pulsed lasers.
X rays can reveal atomic-scale spatial details in liquids and solids because their wavelengths are as short as the distances between atoms. Experiments typically involve aiming an x-ray beam at a sample and measuring the intensity of scattered x rays at each angle around the sample. In so-called inelastic x-ray scattering, researchers also measure the energy of the scattered rays, since x rays sometimes lose energy as they ricochet off of electrons. In theory, the scattering angles lead to nanoscale still pictures, while the energy loss data tell researchers how the pictures change with time. But there is a catch: the mathematical analysis for converting these measurements into still pictures and movies usually involves solving the infamous “phase problem.”
The phase problem is a mathematical one, and it has not been solved for inelastic x-ray scattering in the past. So researchers were left with only indirect views of the motions of electrons. Peter Abbamonte, now at the Brookhaven National Laboratory in Upton, New York, and his colleagues from Cornell University in Ithaca, New York, came up with a solution, and it required lots of high-quality scattering data over a wide range of energies.
So the team set up the experiment at CHESS, the Cornell x-ray facility, and then trekked to the Advanced Photon Source at Argonne National Laboratory in Illinois for a more intense x-ray beam. They aimed the x rays at a container of ordinary water and measured the directions and energies of the scattered radiation. The team then used their new mathematical procedure with this unusually large dataset to create a movie in which each frame covers only 4.13 attoseconds, or seconds. The movie shows how electrons in the water molecules would respond on average if a point of charge were added. The team then decided to shoot a sequel–they imagined firing a gold ion through the water and created a movie showing the “wake” of electron motion that would follow the charged atom.
Abbamonte says that x rays are uniquely suited for these ultrafast measurements, in part because the shortest pulses made with lasers are about 250 attoseconds long. Eric Isaacs of Argonne says the work is “a very cute application of inelastic x-ray scattering.” But he adds that the experiment works particularly well for water, which has few electrons. Extending the technique to other materials with more electrons–such as high-temperature superconductors–may be more challenging.
–Don Monroe
Don Monroe is a freelance science writer in Murray Hill, New Jersey.