Rydberg Atom Takes a Dip in the Cold Sea
Rydberg atoms have at least one valence electron excited to a high-lying state. Immersed in a dense sea of cold atoms, they easily collide with the surrounding atoms and thus can be used to study cold chemical processes. Tilman Pfau from the University of Stuttgart, Germany, Chris Greene from Purdue University, Indiana, and colleagues identified two such processes in a regime in which the atomic sea is extremely dense and cold.
The authors studied a Rydberg atom in an ensemble with a peak density of over neutral atoms per cubic centimeter chilled to a temperature below 1 microkelvin. At these temperatures and densities, all of the atoms move slowly (at roughly a centimeter per second) and some neutral atoms are caught within the orbit of the Rydberg electron. The resulting atomic collisions and chemical processes take place on time and spatial scales that are large enough to identify the processes using spectroscopic techniques.
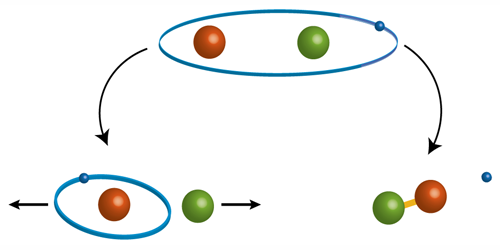
The researchers identified two processes occurring between the Rydberg atom and a neutral atom that limit the Rydberg atom’s lifetime: the formation of a molecule involving the two atoms, and a change to the Rydberg electron’s angular momentum that increases the kinetic energy of the atoms. The odds for these outcomes depend on the principal quantum number n of the Rydberg atom and the ensemble’s density. Remarkably, at the system’s peak density and for n larger than 140, the atom’s lifetime can exceed 10 microseconds even though there are more than 10,000 atoms within the Rydberg electron’s orbit. These long-lived Rydberg states could, for example, be used to image the elusive wave function of Rydberg atoms in real space and in situ within the ensemble.
This research is published in Physical Review X.
–Ana Lopes
Ana Lopes is a Senior Editor of Physics.